Sample Question Paper Class 12 Chemistry 2020-2021 Class: XII Chemistry Session 2020-21 Time: 3 Hours Maximum Marks: 70 General Instructions: Read the following instructions carefully. a) There are 33 questions in this question paper. All questions are compulsory. b) Section A: Q. No. 1 to 16 are objective type questions. Q. No. 1 and 2 are passage based…
Author: Dr. Vikas Jasrotia
General Properties of Group 15 p – Block elements The elements in which the last electron enters in the valence p-sub shell are called the p-block elements. They include elements from groups 13 to 18. Their general electronic configuration is ns2np1-6 where n = 2 (except He which has 1s2 configuration). They, includes metals, non-metals and metalloids. Elements belonging to the s and p-blocks in the periodic table are called the representative elements or main group elements. Inert pair effect: The tendency of ns2 electron pair to participate in a bond formation decreases with the increase in atomic…
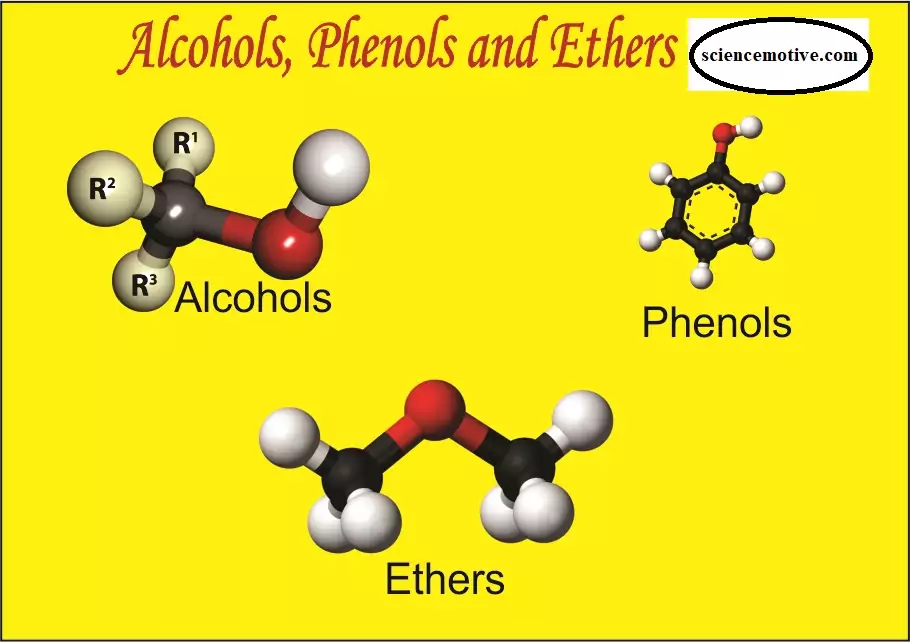
Solved Questions Alcohols, Phenols and Ethers Solved Questions Alcohols, Phenols and Ethers: Que 1. What is the structure and IUPAC name of glycerol? Ans 1. IUPAC name is propane -1,2,3-triol. Que 2. Write the IUPAC name of the following compounds. Ans. (A) 3-Ethyl-5-methylhexan-2,4-diol (B) 1-Methoxy-3-nitrocyclohexan Que 3. Write the IUPAC name of the compound given below Ans 3. 3-Methylpent-2-ene-1,2-diol Que 4. Name the factors responsible for the solubility of alcohols in water. Ans 4. The solubility of alcohols and phenols in water is due to their ability to form hydrogen bonds with water molecules as shown. The solubility decreases with an…
Assertion Reason and Matchup Questions of Amines Assertion Reason and Matchup Questions of Amines: Note: Match the items of Column I and Column II in the following questions 1. Match the reactions given in Column I with the statements given in Column Column I Column II (i) Ammonolysis (a) Amine with a lesser number of carbon atoms (ii) Gabriel phthalimide synthesis (b) Detection test for primary amines. (iii) Hoffmann Bromamide reaction (c) Reaction of phthalimide with KOH and R—X (iv) Carbylamine reaction (d) Reaction of alkyl halides with NH3 Ans 1. (i)-(d) (ii)- (c) (iii)- (a) (iv)- (b) Explanation: (i)…
Solved Subjective and Objective Questions of Amines Solved Subjective and Objective Questions of Amines: Q1. Why Amines are basic in nature? Ans 1. The reaction of amines with mineral acids to form ammonium salts shows that these are basic in nature. Amines have an unshared pair of electrons on nitrogen atom due to which they behave as Lewis base. Q2. In Alkylamines and ammonia which is more basic and why? Ans 2. Due to the electron releasing nature of the alkyl group, it (R) pushes electrons towards nitrogen and thus makes the unshared electron pair more available for sharing with…
Intermolecular Forces and The Gas Laws Intermolecular Forces and The Gas Laws Introduction: Matter is anything that occupies space and has a definite mass. Matter mainly exists in three different states – solid, liquid, and gaseous states. Solids have a definite shape and definite volume. This is because in solids the particles are closely packed and so the intermolecular force of attraction is greater. Liquids have no definite shape but have a definite volume. In liquids, the intermolecular force of attraction is smaller than that in solids. So, the particles do not have a fixed position. Gases have no definite…
Solved Important Questions d & f Block Elements Solved Important Questions d & f Block Elements: Que 1. Cu+ is not stable in an aqueous solution. Why? Ans 1. In an aqueous solution, Cu+ undergoes disproportionation to form a more stable Cu2+ ion. 2Cu+ (aq) → Cu2+ (aq) + Cu(s) The higher stability of Cu2+ in an aqueous solution may be attributed to its greater negative ∆hydH than that of Cu+. It compensates for the second ionization enthalpy of Cu involved in the formation of Cu2+ ions. Que 2. Which is a stronger reducing agent Cr2+ or Fe2+ and why?…
Solved MCQs d & f Block Elements Solved MCQs d & f Block Elements: Que 1. Four successive members of the first-row transition elements are listed below with their atomic numbers. Which one of them is expected to have the highest third ionisation enthalpy? a) Vanadium (Z = 23) b) Chromium (Z = 24) c) Manganese (Z = 25) d) Iron (Z = 26) Ans 1. (c) It can be seen after losing 2 electrons, Mn has 3d5 configuration which is exactly half filled. More energy is required to remove the third electron. Que 2. The aqueous solution containing…
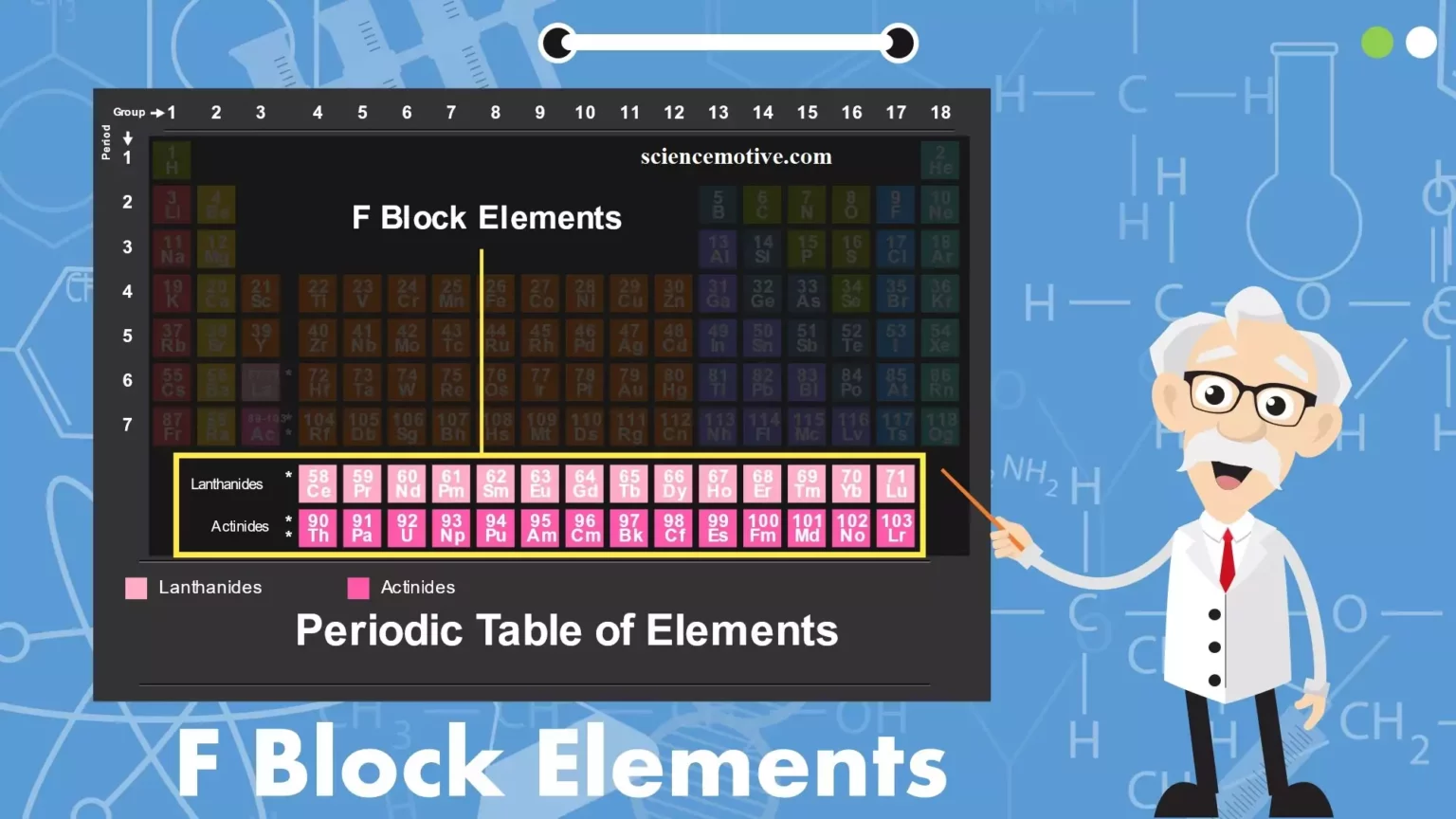
f-Block Elements and Properties f-Block Elements and Properties: The elements in which the last electron enters in the anti-penultimate f-subshell are called f-block elements. They include lanthanides of the 6th period and actinides of the 7th period. The Lanthanoids: Elements’ filling up of 4f – orbitals are called Lanthanoids series. The 14 elements after lanthanum of the 6th period are called lanthanides or lanthanoids or lanthanones or rare earths. They include elements from 58Ce to 71Lu. They are generally represented as Ln. (i) Atomic and Ionic Radii: The overall decrease in atomic and ionic radii from Lanthanum to Lutetium is…
Preparation and Properties of Potassium Permanganate and Potassium Dichromate Preparation and Properties of Potassium Permanganate and Potassium Dichromate: General Properties of First Row Transition Metal Compounds Oxides and oxometal ions: (i) Oxides of metals in low oxidation states + 2 and + 3 (MO, M3O4 and M2O3) are generally basic except Cr2O3 which is amphoteric in character. (ii) Oxides of metals in higher oxidation states + 5 (M2O5, MO3, M2O7) are generally acidic in character. (iii) Oxides of metals in their intermediate oxidation states + 4 (MO2) are generally amphoteric in nature. Besides the oxides, the oxocations, which stabilise V…