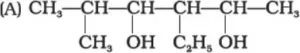Solved Questions Alcohols, Phenols and Ethers
Solved Questions Alcohols, Phenols and Ethers:
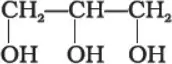
Que 1. What is the structure and IUPAC name of glycerol?
Ans 1. IUPAC name is propane -1,2,3-triol.
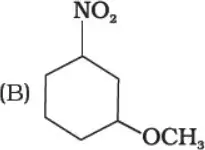
Que 2. Write the IUPAC name of the following compounds.
Ans. (A) 3-Ethyl-5-methylhexan-2,4-diol
(B) 1-Methoxy-3-nitrocyclohexan
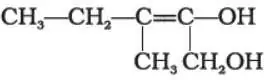
Que 3. Write the IUPAC name of the compound given below
Ans 3. 3-Methylpent-2-ene-1,2-diol
Que 4. Name the factors responsible for the solubility of alcohols in water.
Ans 4. The solubility of alcohols and phenols in water is due to their ability to form hydrogen bonds with water molecules as shown. The solubility decreases with an increase in the size of alkyl/aryl (hydrophobic groups).
Que 5. What is denatured alcohol?
Ans 5. The commercial alcohol is made unfit for drinking by mixing in it some copper sulphate (to give it a colour) and pyridine (a foul-smelling liquid). It is known as the denaturation of alcohol.
Que 6. Suggest a reagent for the following conversion.
Ans 6. Pyridinium chlorochromate (PCC), a complex of chromium trioxide with pyridine and HCl
Que 7. Out of 2-chloroethanol and ethanol which is more acidic and why?
Ans 7. The acidic character of alcohols is due to the polar nature of the O—H bond. 2-Chloroethanol is more acidic due to –I effect of the chlorine atom. It increases the polarity of the O—H bond and increases the acidic strength.
Que 8. Suggest a reagent for conversion of ethanol to ethanal.
Ans 8. A better reagent for oxidation of primary alcohols to aldehydes in good yield is pyridinium chlorochromate (PCC), a complex of chromium trioxide with pyridine and HCl.
Que 9. Suggest a reagent for conversion of ethanol to ethanoic acid.
Ans 9. Strong oxidizing agents such as acidified potassium permanganate are used for getting carboxylic acids from alcohols directly.
Que 9. Out of o-nitrophenol and p-nitrophenol, which is more volatile? Explain.
Ans 9. o-nitrophenol, due to intramolecular hydrogen bonding, is more volatile in nature. In para-nitrophenol, there is intermolecular hydrogen bonding.
Que 10. Out of o-nitrophenol and o-cresol which is more acidic?
Ans 10. In substituted phenols, the presence of electron-withdrawing groups such as nitro group enhances the acidic strength of phenol. This effect is more pronounced when such a group is present at ortho and para positions. It is due to the effective delocalisation of negative charge in phenoxide ion. On the other hand, electron releasing groups, such as alkyl groups, in general, do not favour the formation of phenoxide ion resulting in a decrease in acid strength. Cresols, for example, are less acidic.
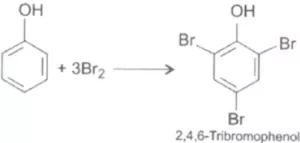
Que 11. When phenol is treated with bromine water, a white precipitate is obtained. Give the structure and the name of the compound formed.
Ans 11. When phenol is treated with bromine water, 2,4,6 –tribromophenol is formed as a white precipitate.
Que 12. Arrange the following compounds in increasing order of acidity and give a suitable explanation. Phenol, o-nitrophenol, o-cresol.
Ans 12. Increasing order of acidity will be o-cresol<phenol< o-nitrophenol.
Explanation: In substituted phenols, the presence of electron-withdrawing groups such as nitro group, enhances the acidic strength of phenol. This effect is more pronounced when such a group is present at ortho and para positions. It is due to the effective delocalization of negative charge in phenoxide ion. On the other hand, electron releasing groups, such as alkyl groups, in general, do not favour the formation of phenoxide ion resulting in a decrease in acid strength. Cresols, for example, are less acidic.
Que 13. Alcohols react with active metals e.g. Na, K etc. to corresponding alkoxides. Write down the decreasing order of reactivity of sodium metal towards primary, secondary and tertiary alcohols.
Ans 13. Decreasing order of reactivity of sodium metal is: 10 > 20 > 30
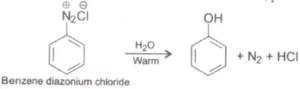
Que 14. What happens when benzene diazonium chloride is heated with water?
Ans 14. Benzene diazonium chloride when heated with water, phenol is
Que 15. Arrange the following compounds in decreasing order of acidity. H2O, ROH, HC ≡ CH.
Ans 15. H2O>ROH>HC ≡ CH
Solved Questions Alcohols, Phenols and Ethers
Que 16. Name the enzymes and write the reactions involved in the preparation of ethanol from sucrose by fermentation.
Ans 16. Ethanol, C2H5OH, is obtained commercially by fermentation, the oldest method is from sugars. The sugar in molasses, sugarcane or fruits such as grapes is converted to glucose and fructose, (both of which have the formula C6H12O6), in the presence of an enzyme, invertase.
C12H22O11 + H2O Invertase C6H12O6 + C6H12O6
Que 17. How can propan-2-one be converted into tert– butyl alcohol?
Ans 17. Using Grignard’s Reagent.
Que 18. Write the structures of the isomers of alcohols with molecular formula C4H10O. Which of these exhibits optical activity?
Ans 18.

The asymmetry of the molecule is responsible for the optical activity in a molecule. In the above structures, it is only butan-2-ol which is asymmetric because of the chiral carbon i.e. the carbon atom attached to four different substituents. Hence, it is optically active.
Que 19. Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
Ans 19. In phenols, the —OH group is attached to sp2 hybridised carbon of an aromatic ring. The carbon-oxygen bond length (136 pm) in phenol is slightly less than that in methanol.
This is due to partial double bond character on account of the conjugation of unshared electron pair of oxygen with the aromatic ring whereas, in alcohols, the oxygen of the — OH group is attached to carbon by a sigma bond formed by the overlap of an sp3 hybridised orbital of carbon with an sp3 hybridised orbital of oxygen. That is why —OH group in phenol is more strongly held as compared to alcohol.
Que 20. Explain why nucleophilic substitution reactions are not very common in phenols.
Ans 20. The —OH group attached to the benzene ring activates it towards electrophilic substitution reaction. Also, it directs the incoming group to ortho and para positions in the ring as these positions become electron-rich due to the resonance effect caused by — OH group. That is why the nucleophilic substitution reaction is not common in phenol.
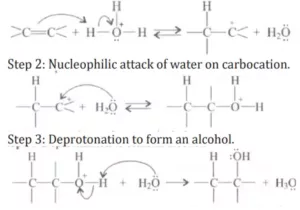
Que 21. Preparation of alcohols from alkenes involves the electrophilic attack on the alkene carbon atom. Explain its mechanism.
Ans 21.
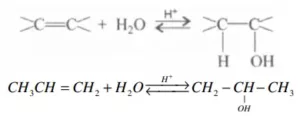
Mechanism: The mechanism of the reaction involves the following three-step:
Step 1: Protonation of alkene to form carbocation by the electrophilic attack of H3O+ H2O + H+ → H3O+
Que 22. Explain why is O=C=O nonpolar while R—O—R is polar.
Ans 22.
Que 23. Why is the reactivity of all the three classes of alcohols with conc? HCl and ZnCl2 (Lucas reagent) different?
Ans 23. Reaction with hydrogen halides: Alcohols react with hydrogen halides to form alkyl halides.
ROH + HX → R—X + H2O
The difference in reactivity of three classes of alcohols with HCl distinguishes them from one another (Lucas test). Alcohols are soluble in Lucas reagent (conc. HCl and ZnCl2) while their halides are immiscible and produce turbidity in solution. In the case of tertiary alcohols, turbidity is produced immediately as they form the halides easily. Primary alcohols do not produce turbidity at room temperature. The reaction is followed by the formation of carbocation since tertiary carbocation is most stable.
Thus, the order of reactivity will be 3° > 2° > 1°.
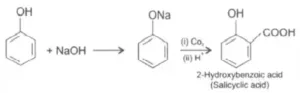
Que 24. Write steps to carry out the conversion of phenol to aspirin.
Ans 24.
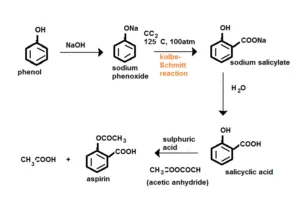
Que 25. Nitration is an example of aromatic electrophilic substitution and its rate depends upon the group already present in the benzene ring. Out of benzene and phenol, which one is more easily nitrated and why?
Ans 25. Phenol will be easily nitrated since the —OH group attached to the benzene ring activates it towards electrophilic substitution. Also, it directs the incoming group to ortho and para positions in the ring as these positions become electron-rich due to the resonance effect caused by —OH group.
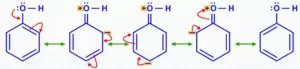
Que 26. In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
Ans 26. Phenoxide ion generated by treating phenol with sodium hydroxide is even more reactive than phenol towards electrophilic aromatic substitution. Hence, it undergoes electrophilic substitution with carbon dioxide, a weak electrophile. Ortho hydroxybenzoic
acid is formed as the main reaction product.
Que 27. The dipole moment of phenol is smaller than that of methanol. Why?
Ans 27. Due to electron-withdrawing effect of the phenyl group, the C—O bond in phenol is less polar, whereas in case of methanol the methyl group has electron releasing effect and hence C— O bond in it is more polar. Dipole moment depends upon the polarity of bonds.
Que 28. Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
Ans 28. In tert-butyl halides, elimination is favoured over substitution, so alkene is the only reaction product and ether is not formed. 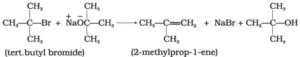
Que 29. Explain why low molecular mass alcohols are soluble in water.
Ans 29. The low molecular mass alcohols are soluble in water due to the presence of intermolecular hydrogen bonding between their molecules. Polar O-H group favours dissolution process whereas non-polar alkyl group does not. As the size of the alkyl group- increases, it overcomes the effect of the polar nature –OH group and the solubility decreases.
Que 30. Explain why p-nitrophenol is more acidic than phenol.
Ans 30. In substituted phenols, the presence of electron-withdrawing groups such as nitro groups enhances the acidic strength of phenol. This effect is more pronounced when such a group is present at ortho and para positions. It is due to the effective delocalisation
of negative charge in phenoxide ion.
Que 31. Explain why alcohols and ethers of comparable molecular mass have different boiling points?
Ans 31. The high boiling points of alcohols are mainly due to the presence of intermolecular hydrogen bonding in them which is lacking in ethers.
Que 32. The carbon-oxygen bond in phenol is slightly stronger than that in methanol. Why?
Ans 32. C—O bond in phenols has partial double bond character due to resonance and hence is difficult to cleave.
Que 33. Arrange water, ethanol and phenol in increasing order of acidity and give a reason for your answer.
Ans 33. phenol>water>ethanol ![]()
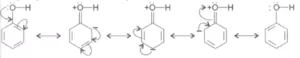
An alkoxide ion is a better proton acceptor than hydroxide ion, which suggests that alkoxides are stronger bases (sodium ethoxide is a stronger base than sodium hydroxide). The reaction of phenol with aqueous sodium hydroxide indicates that phenols are stronger acids than alcohol and water. The ionisation of alcohol and a phenol takes place as follows:
![]()
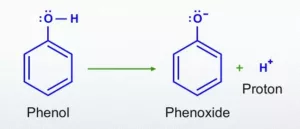
In alkoxide ion, the negative charge is localised on oxygen while in phenoxide ion, the charge is delocalised. The delocalisation of negative charge (structures I-V) makes
Solved Questions Alcohols, Phenols and Ethers