Sample Question Paper Class 12 Chemistry 2020-2021
Class: XII
Chemistry
Session 2020-21
Time: 3 Hours Maximum Marks: 70
General Instructions:
Read the following instructions carefully.
a) There are 33 questions in this question paper. All questions are compulsory.
b) Section A: Q. No. 1 to 16 are objective type questions. Q. No. 1 and 2 are passage based questions carrying 4 marks each while Q. No. 3 to 16 carry 1 mark each.
c) Section B: Q. No. 17 to 25 are short answer questions and carry 2 marks each.
d) Section C: Q. No. 26 to 30 are short answer questions and carry 3 marks each.
e) Section D: Q. No. 31 to 33 are long answer questions carrying 5 marks each.
f) There is no overall choice. However, internal choices have been provided.
g) Use of calculators and log tables is not permitted.
Section A
Sample Question Paper Class 12 Chemistry 2020-2021
Que 1. Read the given passage and answer the questions that follow.
Biomolecules are complex molecules that build up living organisms and required for their growth, maintenance, and ability to reproduce. Carbohydrates are polyhydroxy aldehydes and ketones which are major sources of energy. Monosaccharides are simple sugars that cannot be hydrolyzed. Oligosaccharides, on hydrolysis, gives 2 to 10 molecules of monosaccharides. Polysaccharides like starch and cellulose on hydrolysis give a large number of molecules of glucose a-glucose and b-glucose (Anomers). Proteins are complex nitrogenous polymers of amino acids connected through peptide bonds. The sequence in which amino acids are linked is called the Primary structure. Secondary structures are of 2 types a-helix in globular proteins and b-pleated structure in fibrous proteins involving H-bonds. The tertiary structure has H-bonds, disulphide linkage, ionic bonding, and van der Waals’ forces. Insulin is a hormone for the metabolism of glucose, has a quarternary structure. Denaturation of protein destroys a secondary and tertiary structure, loss of biological activity but primary structure remaining the same. Enzymes are highly specific, work at specific pH, moderate temperature, and catalyze biochemical reactions. Hormones perform specific functions and secreted by endocrine glands. Vitamins are essential for healthy body. A, D, E, K are fat-soluble vitamins. Vitamin C and B1, B2, B6 are water-soluble. B12 is neither water nor fat-soluble. Nucleic acids are polymers of nucleotides. RNA consists of m-RNA, t-RNA, r-RNA. RNA has Adenine, Cytosine, Uracil, and Guanine. It helps in protein synthesis. It cannot replicate. DNA contains deoxyribose, A, C, G, and Thymine. It transfers genetic characteristics. DNA has a double helix structure and undergoes replication.
(a) Name a disaccharide which on hydrolysis give glucose and galactose.
(b) What type of protein is albumin?
(c) Name one non-reducing sugar.
(d) Which one is the complementary base of cytosine in one strand of DNA to that in another strand of DNA?
OR
Which linkage by which nucleotides are joined together between 5′ and 3′ atoms of pentose sugar? 4
Que 2. The solutions which obey Raoult’s law over the entire range of concentration are known as the ideal solution. However, there are many solutions that do not obey Raoult’s law, such solutions are called non-ideal solutions. They show deviation from Raoult’s law i.e. ideal behavior which may be positive or negative. The solution of ethanol and acetone, chloroform and acetone, etc. fall into this category.
(a) State Raoult’s law.
(b) What type of deviation from Raoult’s law is shown by a mixture of ethanol and acetone and why?
(c) What type of deviation from Raoult’s law is shown by a solution of chloroform and acetone and why?
(d) Give two examples of solutions which are nearly ideal in behavior. 4
Questions Nos. 3 -11 are multiple-choice questions carrying 1 mark each
Que 3. Which among the following statement is not true for the rate constant of a reaction?
a) Unit of rate constant depend upon the order of reaction
b) Rate constant depends upon the concentration of the reactants
c) Rate constant has a definite value at a particular temperature
d) Rate constant changes with temperature
Que 4. Which is correct in the case of van der Waal adsorption? High temperature, high pressure
i) Low temperature, high pressure
ii) Low temperature, low pressure
iii) High temperature, low pressure
OR
The physical adsorption is due to
i) van der Waal’s forces
ii) Strong electrostatic forces of attraction
iii) Hydrogen bonding
iv) Covalent bond
Que 5. Among the following, which bivalent ion of the first transition series shows maximum magnetic moment?
i) Co2+
ii) Ni2+
iii) Mn2+
iv) Fe2+
Que 6. Pure N2 is prepared in the laboratory by heating a mixture of:
i) NH4Cl and NaOH
ii) NH4OH and NaCl
iii) NH4Cl and NaNO3
iv) NH4Cl and NaNO2
Que 7. Reactions with iodine in preparation of aryl iodide from arenes require the presence of
i) diazonium salt
ii) an oxidizing agent
iii) a reducing agent
iv) ZnCl2 catalyst
Que 8. Dow’s process involves
i) Nucleophilic substitution
ii) Electrophilic addition
iii) Nucleophilic addition
iv) Electrophilic substitution
OR
Which of the following reactions will not result in the formation of a carbon-carbon bond?
i) Friedel Crafts acylation
ii) Wurtz reaction
iii) Cannizzaro reaction
iv) Reimer-Tieman reaction
Que 9. The correct sequence of steps involved in the mechanism of Cannizzaro’s reaction is
i) electrophilic attack by OH–, transfer of H+ and transfer of H–
ii) the transfer of H+, nucleophilic attack, and transfer of H–
iii) electrophilic attack by OH–, transfer of H+ and transfer of H–
iv) nucleophilic attack, transfer of H– and transfer of H+
OR
A mixture of benzaldehyde and formaldehyde on heating with aqueous NaOH solution gives
i) benzyl alcohol and methyl alcohol
ii) benzyl alcohol and sodium formate
iii) sodium benzoate and methyl alcohol
iv) sodium benzoate and sodium formate
Que 10. If the starting amide has got four carbon atoms and the amine that is formed has got only 3 carbon atoms, then the reaction is called
i) Gabriel synthesis
ii) carbylamines reaction
iii) Hoffmann bromamide reaction
iv) Clemmensen reduction
OR
Identify the correct IUPAC name
(a) (CH3CH2)2NCH3 = N-Ethyl-N-methylethanamine
(b) (CH3)3CNH2 = 2-methylpropan-2-amine
(c) CH3NHCH(CH3)2 = N-Methylpropan-2-amine
(d) (CH3)2CHNH2 = 2, 2-Dimethyl-N-propanamine
Que 11. If 75% of a first-order reaction was completed in 32 min, then 50% of the reaction was completed in
i) 24 min
ii) 4 min
iii) 16 min
iv) 8 min
In the following questions (Q. No. 12 – 16) a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices. Each question is of 1 mark.
(A) Both assertion and reason are correct statements, and the reason is the correct explanation of the assertion.
(B) Both assertion and reason are correct statements, but the reason is not the correct explanation of the assertion.
(C) Assertion is correct, but the reason is the wrong statement.
(D) Assertion is wrong, but the reason is the correct statement.
Que 12. Assertion: The two strands of DNA are complementary to each other
Reason: The hydrogen bonds are formed between specific pairs of bases.
Que 13. Assertion: Both rhombic and monoclinic sulphur exist as S8 but oxygen exists as O2.
Reason: Oxygen forms pπ – pπ multiple bonds due to the small size and small bond length but pπ – pπ bonding is not possible in sulphur.
Que 14. Assertion: KCN reacts with methyl chloride to give methyl isocyanide
Reason: CN– is an ambident nucleophile.
Que 15. Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilization of the phenoxide ion by dispersal of negative charge due to resonance.
Que 16. Assertion: The boiling points of alkyl halides decrease in the order: RI > RBr > RCl > RF
Reason: The boiling points of alkyl chlorides, bromides and iodides are considerably higher than that of the hydrocarbon of comparable molecular mass.
SECTION-B
Sample Question Paper Class 12 Chemistry 2020-2021
Q.No 17 – 25 are Short Answer Type I carrying 2 marks each
Que 17. Explain the following observations:
(i) H2S is less acidic than H2Te.
(ii) Fluorine is a stronger oxidizing agent than chlorine.
OR
Give reasons for the following:
(i) Reducing character decreases from SO2 to TeO2.
(ii)Bond angle decreases from H2O to H2Te.
Que 18. A reaction is of second order with respect to a reactant. How is the rate of reaction affected if the concentration of the reactant is reduced to half? What is the unit of the rate constant for such a reaction?
OR
For the hydrolysis of methyl acetate in an aqueous solution, the following results were obtained:
| Time/s | 0 | 30 | 60 |
| CH3COOCH3 mol/lt | 0.60 | 0.30 | 0.15 |
(i) Show that it follows pseudo-first-order reaction, as the concentration of water remains constant.
(ii) Calculate the average rate of reaction between the time interval 30 to 60 seconds.
(Given log 2 = 0.3010, log 4 = 0.6021)
Que 19. When 1.5 g of a non-volatile solute was dissolved in 90 g of benzene, the boiling point of benzene raised from 353.23 K to 353.93 K. Calculate the molar mass of the solute. (Kb for benzene = 2.52 K kg mol–1)
Que 20. Silver crystallizes in fcc lattice. If the edge length of the cell is 4.07 × 10–8cm and density is 10.5 g cm–3, calculate the atomic mass of silver.
OR
In a compound, nitrogen atoms (N) make cubic close-packed lattice and metal atoms (M) occupy one-third of the tetrahedral voids present. Determine the formula of the compound formed by M and N?
Que 21. Write the state of hybridization, shape, and IUPAC name of the complex [CoF6]3–. (Atomic no. of Co = 27)
Que 22. A greenish-yellow gas ‘A’ with pungent and suffocating odour, is a powerful bleaching agent. ‘A’ on treatment with dry slaked lime it gives bleaching powder. Identify ‘A’ and explain the reason for its bleaching action. Write the balanced chemical equation for the reaction of ‘A’ with hot and concentrated NaOH.
OR
What happens when
(i) conc.H2SO4 is added to Cu?
(ii) SO3is passed through water?
Write the equations also.
Que 23. Why is [NiCl4]2- paramagnetic while [NiCN4]2- is diamagnetic? (Atomic No. of Ni=28) Why is low spin tetrahedral complex rarely observed?
Que 24. A strip of nickel metal is placed in a 1 molar solution of Ni(NO3)2 and a strip of silver metal is placed on a 1-molar solution of AgNO3. An electrochemical cell is created when the two solutions are connected by a salt bridge and the two strips are connected by wires to a voltmeter.
Calculate the cell potential, E, at 250C for the cell if the initial concentration of Ni(NO3)2 is 0.100 molar and the initial concentration of AgNO3 is 1.00 molar. [EoNi2+/Ni = -0.25 V, EoAg+/Ag = -0.80 V, log 10-1 = -1
Que 25. (i) Copper has exceptionally positive E0M2+/ M value .Why?
(ii) Which element in the first transition series is a strong reducing agent in +2 oxidation state and why?
SECTION-C
Sample Question Paper Class 12 Chemistry 2020-2021
Question Nos. 26 -30 are Short Answer Type II carrying 3 marks each.
Que 26. Calculate the freezing point of an aqueous solution containing 60 g of glucose (Molar mass = 180 g mol–1) in 250 g of water. (Kf of water = 1.86 K kg mol–1).
Que 27. (i) Differentiate between Chemisorption and Physiosorption.
(ii) Out of MgCl2 and AlCl3which one is more effective in causing a coagulation of negatively charged sol and why?
(iii) Out of sulphur sol and proteins which one forms multimolecular colloids?
Que 28. (a) Why on dilution the Λm of CH3COOH increases drastically while that of CH3COONa increases gradually?
(b) The conductivity of 0.20 M solution of KCl at 298 K is 0.025 S cm-1. Calculate its molar conductivity.
OR
(a) Value of standard electrode potential for the oxidation of Cl– ions is more positive than that of water, even then in the electrolysis of aqueous sodium chloride, why is Cl– oxidized at anode instead of water?
(b) Calculate Λom for acetic acid. Given that
Λom (HCl) = 426 S cm2mol-1, Λom (NaCl)= 126 S cm2mol-1 Λom (CH3COONa)= 91 S cm2mol-1
Que 29. (a) Complete the following reactions:
(i) Cl2+H2O→ (ii) XeF6+3H2O →
(b)Account for the following observations:
(i) Among the halogens, F2 is the strongest oxidising agent.
(ii) Acidity of oxo-acids of chlorine is HOCl < HOClO < HOClO2 < HOClO3.
OR
Draw the structures of the following molecules:
(i) XeOF4 (ii) H2SO4
(iii) How would you account for the following?
(a)The electron gain enthalpy with negative sign is less for oxygen than that for sulphur.
(b) Fluorine never acts as the central atom in polyatomic interhalogen compounds.
Que 30. i) Illustrate the following reactions giving a chemical equation in each case:
Gabriel phthalimide synthesis OR Hoffmann’s bromamide reaction.
ii) Arrange the following
in an increasing order of their basic strengths:C6H5-NH2 ,CH3-CH2-NH2, CH3-NH-CH3
in an increasing order of solubility in water CH3-NH2, (CH3)3N, CH3, CH3-NH-CH3
iii)Give a chemical test to distinguish between C6H5CH2NH2 and C6H5NH2
SECTION -D
Sample Question Paper Class 12 Chemistry 2020-2021
Question Nos. 31 to 33 are long answer type carrying 5 marks each.
Que 31. (i) An organic compound (A) has a characteristic odour. On treatment with NaOH, it forms compounds (B) and (C). Compound (B) has molecular formula C7H8O which on oxidation gives back (A). The compound (C) is a sodium salt of an acid. When (C) is treated with soda-lime, it yields an aromatic compound (D). Deduce the structures of (A), (B), (C) and (D). Write the sequence of reactions involved.
OR
(i) An organic compound ‘X’ having molecular formula C4H8O gives orange-red ppt. with 2, 4-DNP reagent. It does not reduce Tollens’ reagent but gives yellow ppt. of iodoform on heating with NaOI. Compound X on reduction with LiAlH4 gives compound ‘Y’ which undergoes dehydration reaction on heating with conc. H2SO4 to form but-2-ene. Identify the compounds X and Y and explain the reactions.
(ii) Write the reaction for cross aldol condensation of acetone and ethanol
Que 31. (i) Write the formula of reagents used in the following reactions:
(a) Bromination of phenol to 2, 4, 6-tribromophenol
(b) Hydroboration of propene and then oxidation to propanol.
(ii) Arrange the following compound groups in the increasing order of their property indicated:
(a) p-nitrophenol, ethanol, phenol (acidic character)
(b) Propanol, propane, propanal (boiling point)
(iii) Write the mechanism of the following reaction: Hydration of Alcohol
OR
i) Why is the reactivity of all the three classes of alcohols with conc. HCl and ZnCl2(Lucas reagent) different?
ii) Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain
iii) In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
iv) Why is the C—O—H bond angle in alcohols slightly less than the tetrahedral angle whereas the C—O—C bond angle in ether is slightly greater?
v) Explain why alcohols and ethers of comparable molecular mass have different boiling points?
Que 33. a)How do you convert the following:
(i) Prop-1-ene to 1-fluoropropane
(ii) Chlorobenzene to 2-chlorotoluene
(iii) Ethanol to propane nitrile
b) Give reasons:
(i) C—Cl bond length in chlorobenzene is shorter than C—Cl bond length in CH3—Cl.
(ii) SN1 reactions are accompanied by racemization in optically active alkyl halides
OR
a) Amongst the isomeric alkanes of molecular formula C5H12, identify the one that on photochemical chlorination yields a single monochloride.
b) Write the major products in the following reactions:
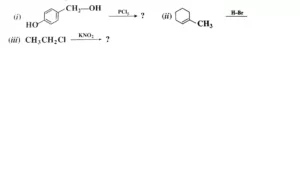
Sample Question Paper Class 12 Chemistry 2020-2021



