Pseudo First Order Reaction Pseudo First Order Reaction: The reactions which are not truly of first order but become reactions of first-order under certain conditions are called pseudo-first-order reactions. Let us take a general reaction A + B → C The rate of reaction depends upon the concentrations of both A and B but one of the components is present in large excess and thus its concentration hardly changes as the reaction proceeds so the rate is independent of that particular component. So, if component B is in large excess and the concentration of B is very high as compared to that…
Author: Dr. Vikas Jasrotia
Chemical Kinetics Chemistry Class 12 Notes -ve sign before ΔR indicates the decrease in the concentration of reactant. Chemical Kinetics Chemistry Class 12 Notes Factors Affecting Rate of Reaction: i). The concentration of reactant ii). Surface area iii). Temperature iv). Nature of reactant v). Presence of catalyst. Dependence rate on Concentration: Rate Law: The rate of reaction is directly proportional to the product of the concentration of reactant and each concentration is raised to some power which may or may not be equal to stereochemistry experimentally. For a general reaction, aA + bB → cC + dD, Rate α [A]x[B]y Rate…
Answers Practice Questions The Solid State Ans 1. AgBr has both schottky and frenkel defects. In general, silver halides show both Schottky and Frenkel defects. It is because AgBr crystal has a coordination number of 6 which is neither high nor low. The radius ratio of AgBr is also intermediate, thus AgBr satisfies the conditions to exhibit both types of defects. Ans 2. (a). Packing Efficiency of Simple Cubic Unit Cell: (b). Packing Efficiency of Body Centred Unit Cell: (c). Packing Efficiency in Face Centred Unit cell: Ans 3. The smallest repeating unit of the space lattice is Unit…
Practice Questions The Solid State The Solid State Que 1. Write the name of a crystal showing both Schottky and Frenkel defects. Que 2. Which of the following lattices has the highest packing efficiency? Explain your answer: (a) Simple Cubic (b) Body Centred Cubic (c) Face Centred Cubic Que 3. The smallest repeating unit of the space lattice is _______ Que 4. A compound is formed by two elements P and Q. Atoms of ‘Q’ (anion) make hcp lattice and those of ‘P’ (cation) occupy all the tetrahedral voids. What is the formula of the compound? Practice…
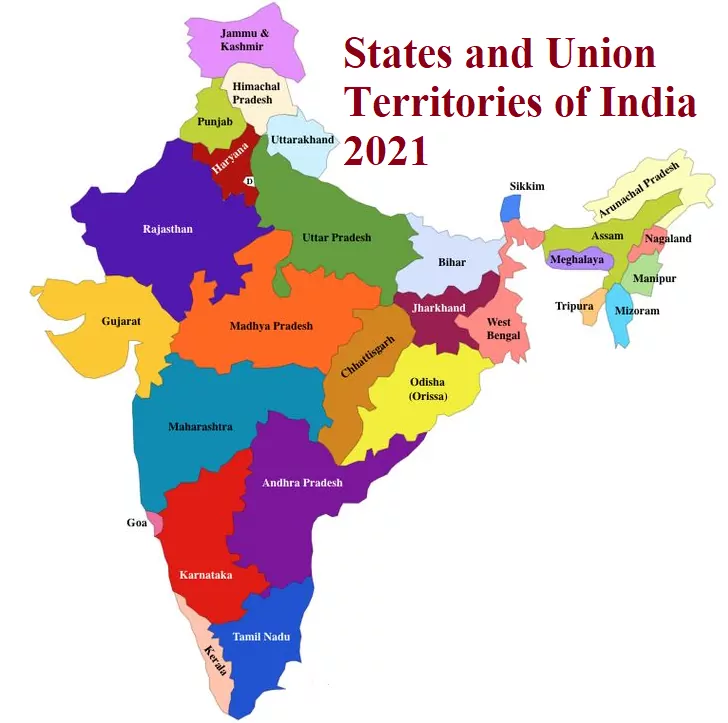
States and Union Territories of India 2021 States and Union Territories of India 2021 S.No States Name Capital Founded on Area (Km2) 1 Andhra Pradesh Hyderabad (Proposed Capital Amaravati) 1 Nov. 1956 160,205 2 Arunachal Pradesh Itanagar 20 Feb. 1987 83,743 3 Assam Dispur 26 Jan. 1950 78,438 4 Bihar Patna 26 Jan. 1950 94,163 5 Chhattisgarh Raipur 1 Nov. 2000 135,191 6 Goa Panaji 30 May. 1987 3,702 7 Gujarat Gandhinagar 1 May. 1960 196,024 8 Haryana Chandigarh 1 Nov. 1966 44,212 9 Himachal Pradesh Shimla 25 Jan. 1971 55,673 10 Jharkhand Ranchi 15 Nov. 2000 79714 11 Karnataka…
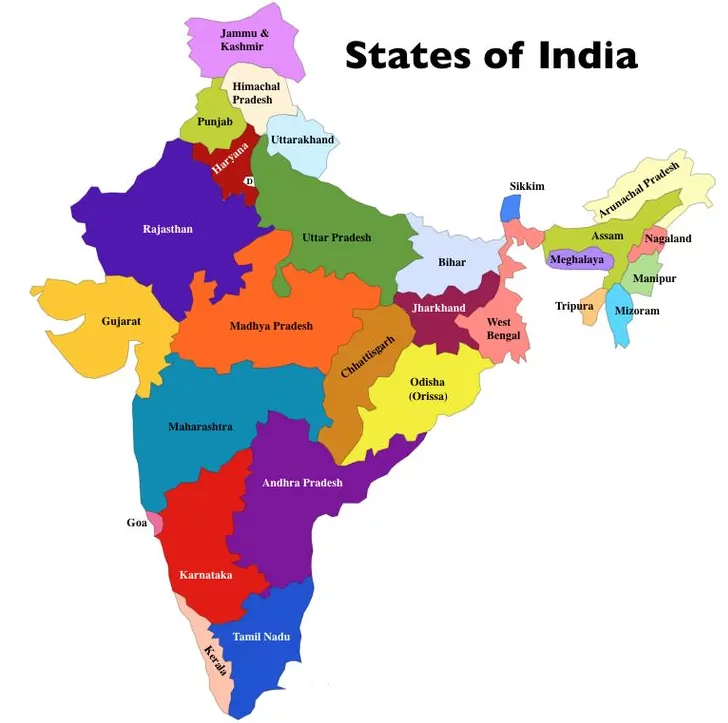
States and Capitals of India 2021 States and Capitals of India 2021 S.No States Name Capital Founded on Area (Km2) 1 Andhra Pradesh Hyderabad (Proposed Capital Amaravati) 1 Nov. 1956 160,205 2 Arunachal Pradesh Itanagar 20 Feb. 1987 83,743 3 Assam Dispur 26 Jan. 1950 78,438 4 Bihar Patna 26 Jan. 1950 94,163 5 Chhattisgarh Raipur 1 Nov. 2000 135,191 6 Goa Panaji 30 May. 1987 3,702 7 Gujarat Gandhinagar 1 May. 1960 196,024 8 Haryana Chandigarh 1 Nov. 1966 44,212 9 Himachal Pradesh Shimla 25 Jan. 1971 55,673 10 Jharkhand Ranchi 15 Nov. 2000 79714 11 Karnataka Bengaluru(Formerly Bangalore)…
Electrochemistry Class XII Solved Numericals Que 1. 0.05 M NaOH solutions offered resistance of 31.16 Ω in a conductivity cell at 298 K. If cell constant is 0.367 cm–1. Find out the molar conductivity of NaOH solution. Ans 1. Given Molarity = 0.05 M Cell Constant = 0.367 cm-1 Que 2. Consider the following cell reaction: 2Fe(s) + O2 (g) + 4H+ (aq) → 2Fe2+ (aq) + 2H2O(l); E0 = 1.67V. At [Fe2+] = 10–3 M, p(O2) = 0.1 atm and pH = 3. What will be the cell potential at 250C? Ans 2. Here n = 4, and [H+]…
Electrochemistry Important questions for Board Que 1. i) Define the molar conductivity of a solution and explain how molar conductivity changes with a change in concentration of solution for a weak and strong electrolyte. ii) The resistance of a conductivity cell containing 0.001M KCl solution at 298 K is 1500Ω. What is the cell constant, if the conductivity of 0.01 M KCl solution at 298 K is 0.146 × 10–3 S cm–1? Ans 1. i) Molar conductivity – it is defined as the conductance of the solution which contains one mole of an electrolyte such that the entire solution…
Electrochemistry Neet Important Solved Questions Que 1. Which of the following statements is incorrect regarding electrochemistry? (a) It is the study of the production of electricity from the energy released during spontaneous chemical reactions. (b) NaOH, Cl2, alkali, and alkaline earth metals are prepared by electrochemical methods. (c) The demerit associated with electrochemical methods is that they are more polluting. Thus they are eco[1]destructive. (d) Electrochemical reactions are more energy-efficient and less polluting. Ans 1. (c) Electrochemical reactions are more energy-efficient and less polluting. Thus the study of electrochemistry is important to create new eco-friendly technologies. Que 2. For a…
Solutions Chemistry Important NEET Questions Que 1. How many grams of concentrated nitric acid solution should be used to prepare 250 mL of 2.0 M HNO3? The concentrated acid is 70% HNO3. (a) 70.0 g conc. HNO3 (b) 54.0 g conc. HNO3 (c) 45.0 g conc. HNO3 (d) 90.0 g conc. HNO3 Ans 1. (c) Explanation: Que 2. In water-saturated air, the mole fraction of water vapour is 0.02. If the total pressure of the saturated air is 1.2 atm, the partial pressure of dry air is (a) 1.18 atm (b) 1.76 atm (c) 1.176 atm (d) 0.98 atm.…