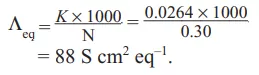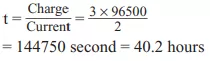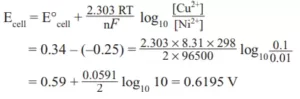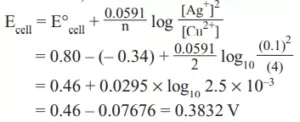Electrochemistry Class XII Solved Numericals
Que 1. 0.05 M NaOH solutions offered resistance of 31.16 Ω in a conductivity cell at 298 K. If cell constant is 0.367 cm–1. Find out the molar conductivity of NaOH solution.
Ans 1. Given Molarity = 0.05 M
Cell Constant = 0.367 cm-1
Que 2. Consider the following cell reaction:
2Fe(s) + O2 (g) + 4H+ (aq) → 2Fe2+ (aq) + 2H2O(l); E0 = 1.67V. At [Fe2+] = 10–3 M, p(O2) = 0.1 atm and pH = 3. What will be the cell potential at 250C?
Ans 2. Here n = 4, and [H+] = 10– 3 (as pH = 3)
Applying Nernst equation
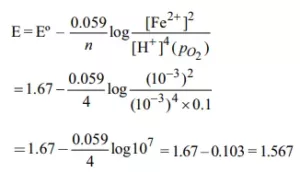
Que 3. A salt solution of 0.30 N placed in a cell whose electrodes are 1.9 cm apart and 3.6 cm2 in area offers a resistance of 20 Ω. Calculate equivalent conductivity of solution.
Ans 3. Resistance (R) = 20 Ω
Length (l) = 1.9 cm a = 3.6 cm2 , Normality (N) = 0.30 N
Cell constant = l/a = 1.9/3.6 = 0.528 cm-1
Specific conductance (K) = Conductance × Cell constant = (1/R) × cell constant
= (1/20) × 0.528 = 0.0264 S cm–1
Electrochemistry Class XII Solved Numericals
Que 4. The electrode potential E (Zn2+/Zn) of a zinc electrode at 25°C with an aqueous solution of 0.1 M ZnSO4 is.
Ans 4.

Que 5. The molar conductivities of CH3COOH at 25°C at the concentration of 0.1 M and 0.001 M are 5.20 and 49.2 cm2 mol–1 respectively. Calculate the degree of dissociation of CH3COOH at these concentrations. (Λ∞m (CH3COOH) = 390.7 S cm2 mol–1).
Ans 5.
Que 6. If 0.01 M solution of an electrolyte has a resistance of 40 ohms in a cell having a cell constant of 0.4 cm–1, then find its molar conductance in ohm–1 cm2 mol–1.
Ans 6. Molarity = 0.01 M
Resistance = 40 ohm
Cell constant l/A = 0.4cm-1.
Electrochemistry Class XII Solved Numericals
Que 7. If Λ∞m of HCl, NaCl, and CH3COONa is 425, 128, and 96 Ω–1 cm2 mol–1 respectively, calculate the value of Λ∞m for acetic acid.
Ans 7. Λ∞m CH3COOH = Λ∞m CH3COONa + Λ∞m HCl – Λ∞m NaCl
= 96.0 + 425.0 – 128.0 = 393.0 Ω–1 cm2 mol–1.
Que 8.
| Electrolyte | KCl | KNO3 | HCl | NaOAc | NaCl |
| Λ∞ (Scm2 mol–1) | 149.9 | 145 | 426.2 | 91 | 126.5 |
Calculate Λ∞HOAc using appropriate molar conductance of the electrolytes listed above at infinite dilution in H2O at 25°C.
Ans 8. Λ∞Hcl = 426.2 ………………(1)
Λ∞AcONa = 91.0 …………………………….(2)
Λ∞NaCl = 126.5 ………………………….….(3)
Λ∞HOAc = (1) + (2) + (3)
= [426.2 + 91.0 + 126.5] = 390.7 Scm2 mol–1
Que 9. How many hours does it take to reduce 3 moles of Fe3+ to Fe2+ with the 2.0-ampere current? (F= 96500 C mol–1).
Ans 9. Fe3+ + e– → Fe2+
Electrochemistry Class XII Solved Numericals
Que 10. At 25°C, the molar conductance at infinite dilution for the strong electrolytes NaOH, NaCl, and BaCl2 is 248 × 10–4, 126 × 10–4 and 280 × 10–4 Sm2mol–1 respectively. Find Λ0m Ba(OH)2 in S m2mol–1.
Ans 10.
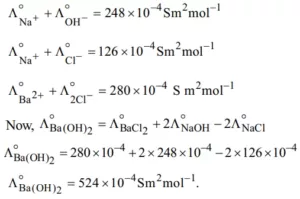
Que 11. How many grams of silver can be plated out on a serving tray by electrolysis of a solution containing silver in +1 oxidation state for a period of 8.0 hours at a current of 8.46 amperes? What is the area of the tray if the thickness of the silver plating is 0.00254 cm? The density of silver is 10.5 g/cm3.
Ans 11.
Que 12. Find the number of coulombs required to reduce 12.3 g of nitrobenzene to aniline.
Ans 12. As per the equation
Electrochemistry Class XII Solved Numericals
Que 13. Calculate the quantity of electricity required to reduce 6.15 g of nitrobenzene to aniline if the current efficiency is 68%. If potential drops across the cell is 7.0 volts. Calculate the energy consumed in the process.
Ans 13. C6H5NO2 + 6H+ + 6e– → C6H5NH2 + 2H2O

Que 14. Calculate the cell potential of the given cell at 25°C. (R = 8.31 J K–1 mol–1, F = 96500 C mol–1).
Ni (s)|Ni2+(0.01 M) || Cu2+ (0.1 M)|Cu(s) Given: E0Cu2+/Cu = + 0.34 V E0 (Ni2+/Ni) = – 0.25 V.
Ans 14.
Que 15. Calculate the cell potential for the cell containing 0.10 M Ag+ and 4.0 M Cu2+ at 298 K. Given E0Ag+ /Ag = 0.80 V, E0Cu2+/Cu = 0.34 V
Ans 15. Cu (s)|Cu2+ (4.0 M) || Ag+ (0.1 M)|Ag (s) Here n = 2
Electrochemistry Class XII Solved Numericals