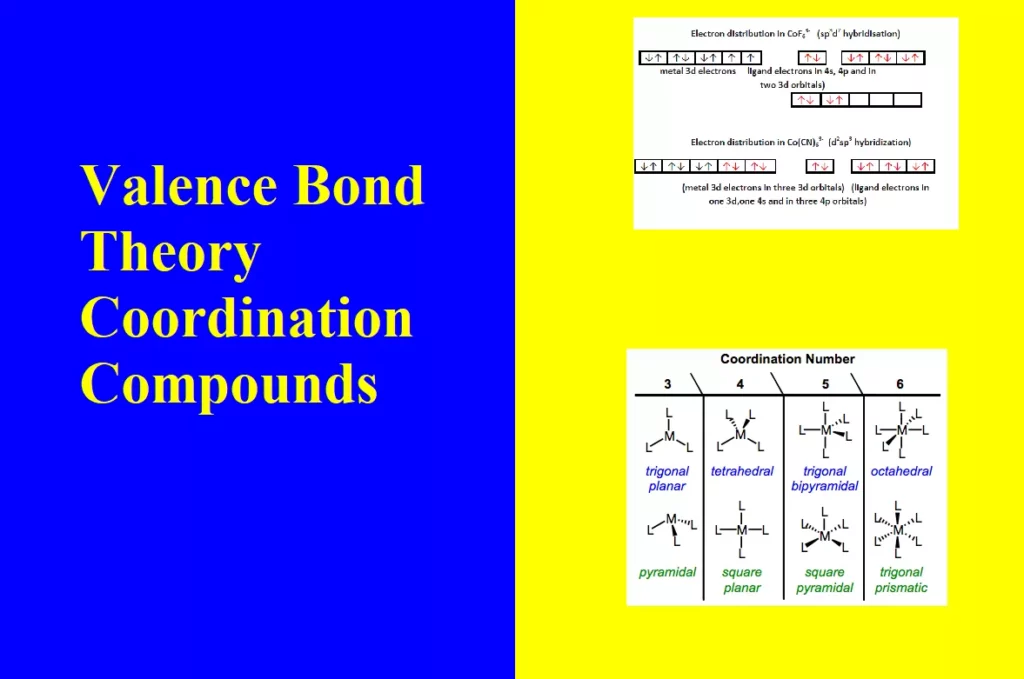
Valence Bond Theory Coordination Compounds Valence Bond Theory The bonding in coordination compounds can be explained by Valence Bond Theory (VBT). It was developed by Pauling. This theory mainly deals with the electronic structure of central metal ion, geometry, magnetic properties of complex, kind of bonding. According to this theory, the metal atom or ion under the influence of ligands form inner orbital and outer orbital complex. These hybridized orbitals are allowed to overlap with ligand orbitals that can donate electron pairs for bonding. The main features of the valence bond theory are summarized below: 1. Inner orbital complex: When…
Author: Dr. Vikas Jasrotia
Coordination Compounds Class 12 Important Questions Que 1. Identify the complexion and counter ion in K4[Fe(CN)6]. Ans 1. [Fe(CN)6]4– and K+ respectively. Que 2. What is ambidentate ligand? Give one example. Ans 2. Monodentate ligands contain more than one coordinating atom. Example, CN−. Que 3. How many geometrical isomers are possible in the following coordination entities: [Cr(C2O4)3]3– [CoCI3(NH3)3]. Ans 3. (i) C2O4 is a bidentate ligand. So no geometrical isomers possible. (ii) Two two geometrical isomers are possible (fac‐and mer‐isomer). Coordination Compounds Class 12 Important Questions Que 4. What is meant by the denticity of a ligand? Give…
Isomerism in Coordination Compounds Class 12 Isomerism Two or more chemical compounds which have identical chemical formula but different structures are known as isomers and the phenomenon is known as isomerism. The isomers have different arrangements of ligands around the central metal atom. The isomerism shown by co-ordination compounds are broadly divided into two: Structural Isomerism and Stereo Isomerism. Structural Isomerism: These are isomers that differ in the structural arrangement of ligands around the central atom. They are of four types: (a) Ionisation Isomerism (b) Coordinate Isomerism (c) Linkage Isomerism (d) Hydrate Isomerism Isomerism in Coordination Compounds Class 12 (a)…
Nomenclature of Coordination Compounds Class 12 Werner’s Theory of Coordination Compounds Alfred Werner, a Swiss chemist proposed a theory about the nature of bonding in the coordination compounds (complexes) in the year 1893. The main postulates of this theory are: 1. Metals possess two types of linkages (valencies). a) Primary or principal or ionizable links (valency) which are the same as the oxidation state of the metal. b) Secondary or non-ionizable links (valency) which are the same as the coordination number of the central metal atom/ion. This number is fixed for a metal. 2. A metal atom satisfies both primary…
Coordination Compounds Important Topics The concept of coordination compounds originates from the tendency for the complex formation of the transition elements. Complex compounds or coordination compounds are those molecular compounds that retain their identity in solid and in solution are known as complex compounds. K4[Fe(CN)6 ] + H2O → 4K+(aq) + [Fe(CN)6]4− (aq) Molecular or Addition Compounds: When solutions of two or more simple stable compounds in molecular proportion are allowed to evaporate, crystals of new substances, called molecular or addition compounds, are obtained. For Example: CuSO4 + 4NH3 → CuSO4·4NH3 AgCN + KCN → KCN·AgCN There are two…
Important Solved Questions on Mole Concept Que 1. Calculate the number of atoms of each type in 5.3 gram of sodium carbonate (Na2CO3).\ Ans 1. Given mass of sodium carbonate (Na2CO3) = 5.3 g Molar mass of sodium carbonate = 23 × 2 + 12 + 16 × 3 = 106g/mol Number of moles = given mass/ Molar mass = 5.3/106 = 0.05 mol Now, 1 mole of Na2CO3 contains = 2 × 6.022 × 1023 sodium atoms ∴0.05 of Na2CO3 contains = 0.05 × 2 × 6.022 × 1023 sodium atoms = 6.022 × 1022 = 6.022 × 1022 Na atoms. 1 mol of Na2CO3 contains…
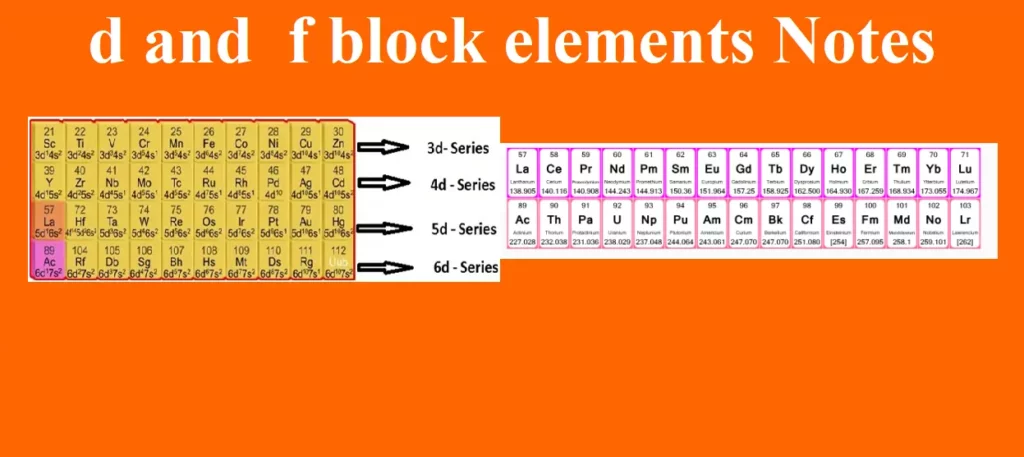
d and f block elements Notes Introduction The elements lying in the middle of the periodic table belonging to groups 3 to 12 are known as d – block elements. The elements in which the last electron enters into the d-orbitals of the penultimate shell i.e. (n–1) d where n is the last shell are called d-block elements. They are placed in between s-block and p-block elements. They show a regular transition from the highly electropositive metals of s-block elements to the less electropositive p-block elements. So, they are called transition elements. Transition elements A transition element is defined as…
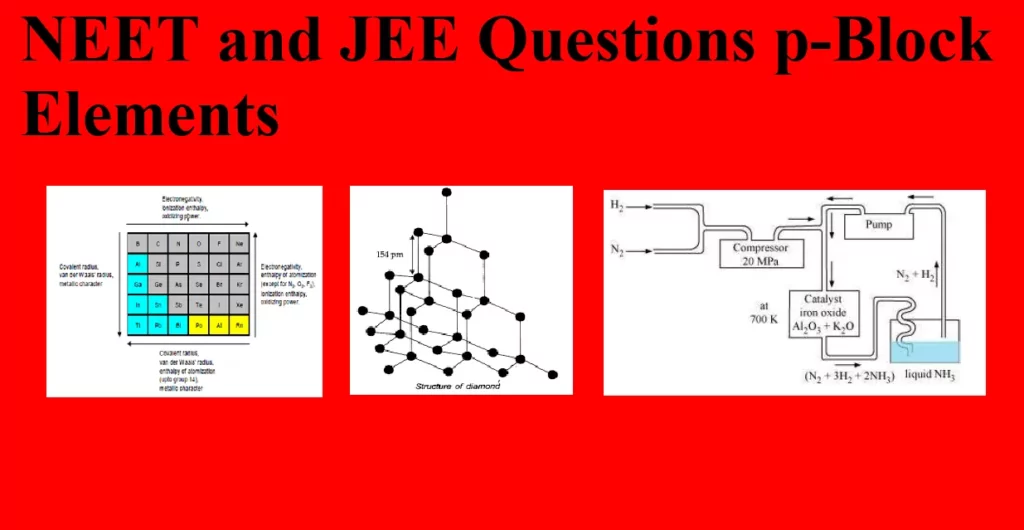
NEET and JEE Questions p-Block Elements Que 1. Which of the following elements does not form stable diatomic molecules. (a) Iodine (b) Phosphorus (c) Nitrogen (d) Oxygen Ans 1. (b) Phosphorus Explanation: Phosphorous forms tetra atomic molecule it does not form stable diatomic molecule whereas the other given elements form stable diatomic molecules(Due to larger atomic size P does not form pi bonds and so it exists as a tetra-atomic molecule in which each P atom is bonded with 3 other P atoms by 3 sigma bond. But, due to smaller atomic size N forms 1 sigma and…
Classification Of Elements And Periodicity In Properties Classification and Blocks in Periodic Table: Topics covered: Earlier classifications Dobereiner’s Triads Newland’s law of octaves Mendeleev’s Periodic Classification Modern Periodic Table or Moseley’s Periodic Law With the discovery of a large number of elements, it became difficult to study the elements individually, so the classification of elements was done to make the study easier. Earlier Classification: Dobereiner’s Triads: In triads, the atomic mass of the middle element is approximately the average of the other two elements. This is known as the Law of Triads. This classification was applicable to very few elements…
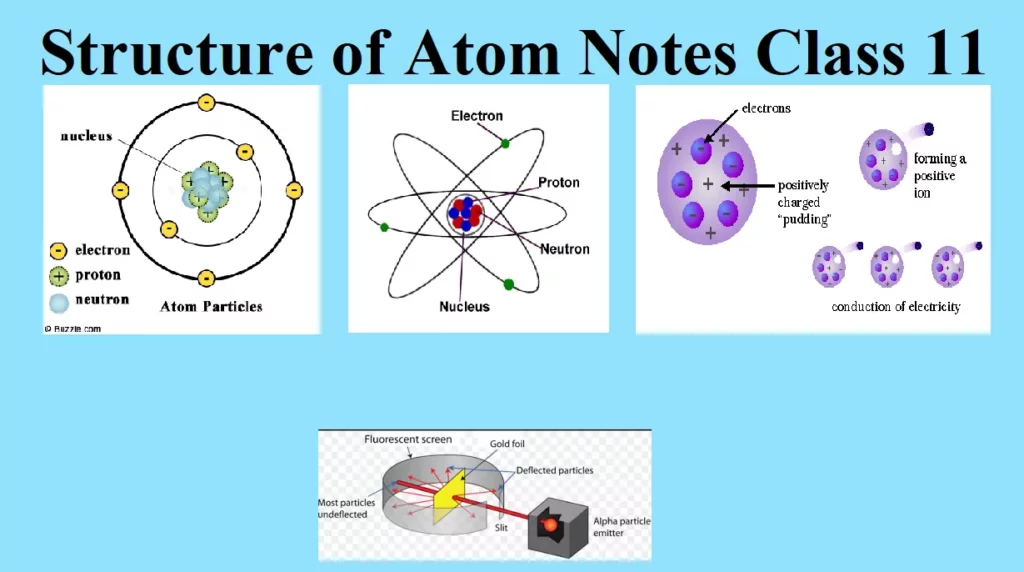
Structure of Atom Notes Class 11 STRUCTURE OF ATOM Structure of Atom The term atom was introduced by Dalton and is defined as the smallest particle of an element that retains all its properties and identity during a chemical reaction. SUB – ATOMIC PARTICLE (i) Discovery of Electron Electron was discovered by J J Thomson by Cathode ray discharge tube experiment. A cathode-ray tube is made of glass containing two thin pieces of metal (called electrodes) sealed in it. The electrical discharge through the gases could be observed only at very low pressures and at very high voltages. When…