Carbon and Its Compounds Extra Questions
Carbon and Its Compounds Extra Questions
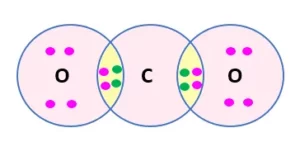
Que 1. What would be the electron dot structure of carbon dioxide which has the formula CO2?
Ans 1. The electron dot structure of Carbon Dioxide (CO2) is
Que 2. Why the bottom of the vessel is getting blackened on the outside, during cooking? Explain.
Ans 2. The bottom of the vessel is getting blackened on the outside because there is not enough oxygen to react with the carbon to produce carbon dioxide, then the unburnt carbon particles are left behind in the form of black particles known as soot. These soot particles stick to the bottom of the vessel making it black.
Que 3. What is a hydrocarbon?
Ans 3. It is a compound of hydrogen and carbon.
Que 4. What are The physical properties of Carbon compounds or covalent compounds?
Ans 4. The covalent compound exhibits the following properties:
(i) These have low melting and boiling point.
(ii) These do not conduct electricity either in the solid-state or in the molten state or in the solution form. They are non-electrolyte.
(iii) These are readily soluble in non-polar solvents like benzene, carbon tetrachloride, etc., but do not dissolve in polar solvents like water.
(iv) The covalent bond is directional in nature.
Que 5. What are the essential constituents of all organic compounds?
Ans 5. Carbon and hydrogen are the essential constituents of all organic compounds. However, carbon tetrachloride (CCl4) is an exception.
Que 6. Write the electron dot structure of the ethene molecule, C2H4.
Ans 6. Electron dot structure of ethane molecule, C2H4
Que 7. What is the role of concentrated H2SO4 in the esterification reaction?
Ans 7. In an esterification reaction, a carboxylic acid reacts with alcohol to form ester and water in the presence of concentrated sulphuric acid. This reaction is reversible and this reverse reaction is called ester hydrolysis. Concentrated sulphuric acid being a strong dehydrating agent removes water from the reaction mixture. As a result, the reaction proceeds only in the forward direction to form an ester.
RCOOH + ROH —-Conc. H2SO4 → RCOOR + H2O
Que 8. What is meant by denatured alcohol? What is the need to denature alcohol?
Ans 8. Ethyl alcohol which contains a small amount of methyl alcohol or copper sulphate is called denatured alcohol. The purpose of denaturing the alcohol is to make it unfit for drinking purposes. Denatured alcohol is also used for industrial purposes.
Que 9. Why does graphite conducts electricity, but not a diamond?
Ans 9. In the case of a diamond, each carbon atom of a single crystal is surrounded by four other carbon atoms by covalent bonds such that they form four corners of a regular tetrahedron. Because of four covalent bonds with each carbon atom, there are no free electrons available. Due to the non-availability of free electrons within the crystalline structure, the diamond acts as a bad conductor of electricity.
In the case of graphite, every carbon atom in a single crystal is covalently bonded to three carbon atoms. As each carbon atom has four valence electrons one valence electron is left free for each carbon atom. These free electrons can be easily made to flow within the crystalline structure of graphite by applying electric potential. Thus, graphite is a good conductor of electricity.
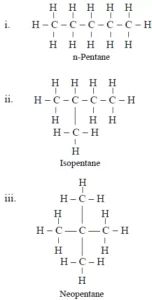
Que 10. How many structural isomers can you draw for pentane?
Ans 10. There are three structural isomers that are possible for pentane.
Que 11. What is catenation?
Ans 11. Carbon has the unique ability to form bonds with the other atoms of carbon which gives rise to large molecules. This property of self linking is called catenation.
Que 12. Covalent compounds have generally low melting points. Why?
Ans 12. Covalent compounds have strong bonds within the molecule and intermolecular forces are very weak and so have low melting and boiling points.
Que 13. Why is candle flame generally yellow?
Ans 13. A candle flame is generally yellow due to the presence of unburnt carbon particles. When light falls on these particles, they scatter yellow color. This shows that the combustion of hydrocarbons present in wax or candle is not complete.
Que 14. Name the functional group present in each of the following organic compounds:
(i) C2H5Cl
(ii) C2H5OH
Ans 14. The functional group present in the compounds are
(i) C2H5Cl – Chloro (halide)
(ii) C2H5OH – Alcohol
Que 15. Give names of the following
(a) An aldehyde derived from ethane
(b) Ketone derived from butane
(c) Compound obtained by the oxidation of ethanol by chromic anhydride
Ans 15. (a) Ethanal (CH3CHO)
(b) Butanone (CH3COCH2CH3)
(c) Ethanal (CH3CHO)
Carbon and Its Compounds Extra Questions
Que 16. Write chemical equations of the reactions of ethanoic acid with
a) Sodium
b) Sodium carbonate
c) Ethanol in the presence of conc. H2SO4
Ans 16. (a) 2CH3COOH + 2Na → 2CH3COONa + H2
(b) 2CH3COOH + Na2CO3 → 2CH3COONa + CO2 + H2O
(c) CH3COOH + C2H5OH —-Conc. H2SO4–→ CH3COOC2H5
Que 17. Why common salt is added in soap making?
Ans 17. Common salt is added to the mixture to make the soap come out of the solution. Though most of the soap separates out on its own but some of it remains in solution. Common salt is added to precipitate out all the soap from the aqueous solution. Actually, when we add common salt to the solution, then the solubility of soap present in it decreases, due to which all the soap separates out from the solution in the form of a solid.
Que 18. What are the two properties of carbon which lead to the huge number of carbon compounds we see around us?
Ans 18. The two features of carbon that give rise to a large number of compounds are as follows:
(i) Catenation − It is the ability to form bonds with other atoms of carbon.
(ii) Tetravalency − With the valency of four, carbon is capable of bonding with four other atoms.
Que 19. Name two allotropes of carbon.
Ans 19. Two allotropes are – Crystalline and amorphous
Crystalline form – Diamond and graphite.
Amorphous form – Charcoal, coal, coke.
Que 20. What is meant by double bond or double covalent bond? Give examples.
Ans 20. The bond between two atoms formed by sharing of two pairs of electrons is called a double bond. Two horizontal lines between two atoms denote a double bond example O=O (O₂)
Carbon and Its Compounds Extra Questions
Que 21. The formula of a hydrocarbon is CnH2n. Name the family to which it belongs and also predict its nature.
Ans 21. The hydrocarbon belongs to the alkene family. It is unsaturated in nature.
Examples:
(i) Formation of Oxygen atom has six electrons in its outermost shell, two short of forming a complete octet. Thus, each oxygen atom in O₂ shares 2 electrons with the other oxygen atom to attain a stable configuration.
(ii) Formation of ethylene molecule (C₂H₄): In ethylene two carbon atom shares two pair of electrons between them and the rest two electrons on each carbon atom share one electron each with two hydrogen atoms. Thus, ethylene molecules have one double bond(C=C) and four single bonds (C-H).
Que 22. Which class of carbon compounds is responsible for the depletion of the ozone layer at the higher level of the atmosphere?
Ans 22. The depletion of the ozone layer is due to the use of chemicals called chlorofluorocarbons.
Que 23. What is a homologous series? State any two characteristics of homologous series?
Ans 23. A series of similarly constituted compounds in which the members present have the same functional group, same chemical properties, and any two successive members is a particular series differ in their molecular formula by CH2 group.
(i) Same functional group
(ii) Same chemical properties.
Que 24. Given a chemical test to distinguish between
(a) Ethene and ethane
(b). Ethanol and ethanoic acid
(c). Soaps and Detergents
Ans 24. (a) Ethene decolorizes the yellow color of bromine while ethane does not.
(b) Ethanoic acid gives a brisk effervescence with sodium hydrogen carbonate while ethanol does not.
(c) Soaps form curdy white precipitate or scum with hard water while detergents do not form any precipitate.
Que 25. What change has been made in the composition of detergents to make them biodegradable?
Ans 25. Detergents made from long-chain hydrocarbons having the minimum branching in their molecules are degraded more easily.
Que 26. Would you be able to check if water is hard by using a detergent?
Ans 26. Detergents are ammonium or sulphonate salts of long-chain carboxylic acids. Unlike soap, they do not react with calcium and magnesium ions present in hard water to form scum. They give a good amount of lather irrespective of whether the water is hard or soft. This means that detergents can be used in both soft and hard water. Therefore, it cannot be used to check whether the water is hard or not.
Que 27. Give two properties of ethanol.
Ans 27. (a) Liquid at room temperature
(b) Soluble in H2O in all proportions
Que 28. Write the general formula for alkanes, alkanes, and alkynes.
Ans 28. The general formula for alkanes CₙH₂ₙ₊₂. Since an alkene contains two hydrogen atoms less than the corresponding alkane, therefore the general formula for alkenes is CₙH₂ₙ. Further, since an alkyne contains 2 hydrogen atoms less than the corresponding alkene hence the general formula for alkynes is CₙH₂ₙ₋₂.
Que 29. Which organic compound is added to make ethanol unfit for drinking purposes? What is the name of the mixture formed?
Ans 29. Methanol which is highly poisonous is added in a small amount to ethanol in order to make it unfit for drinking purposes. The mixture is called methylated spirit or denatured alcohol.
Que 30. Write chemical equations to show what happens when
(i) Ethanol is heated with concentrated sulphuric acid at 443 K.
(ii) Ethanol reacts with ethanoic acid in the presence of an acid acting as a catalyst.
(iii) An ester reacts with a base.
Ans 30. (i) When ethanol is heated with concentrated sulphuric acid at 443K, it gets dehydrated to form ethene
CH3CH2OH + H2SO4(Conc.) → CH2=CH2 + H2O
(ii) When ethanol reacts with ethanoic acid on warming in the presence of a few drops of conc. sulphuric acid to form a sweet-smelling ester, ethyl ethanoate
CH3COOH + C2H5OH —-Conc. H2SO4–→ CH3COOC2H5
(iii) When an ester is heated with a base (sodium hydroxide) solution then the ester gets hydrolyzed (breaks down) to form the parent alcohol and sodium salt of the carboxylic acid.
CH3COOC2H5 + NaOH → CH3COONa + C2H5OH
Carbon and Its Compounds Extra Questions