Assignment on Redox reaction (Unsolved) Assignment on Redox reaction (Unsolved) Q.1. The colorless solution of silver nitrate slowly turns brown on adding copper chips to it because of (a) Dissolution of Copper (b) Oxidation of Ag+ → Ag (c) Reduction of Cu2+ ions (d) Oxidation of Cu atoms. Q.2. The oxidation process involves (a) Increase in oxidation number (b) Decrease in oxidation number (c) No change in oxidation number (d) none of the above Q.3. The oxidation number of Mn is maximum in (a) MnO2 (b) K2MnO4 (c) Mn3O4 (d) KMnO4. Q.4. KMnO4reacts with oxalic acid according to the…
Author: Dr. Vikas Jasrotia
Alcohol Phenol and Ether MCQ for NEET pdf Alcohol Phenol and Ether MCQ for NEET pdf Que 1. Arrange the following compounds in increasing order of boiling point. Propan-1-ol, butan-1-ol, butan-2-ol, pentan-1-ol: (a) Propan-1-ol, butan-1-ol, butan-2-ol, pentan-1-ol (b) Pentan-1-ol, butan-2-ol, butan-1-ol, propan-1-ol (c) Pentan-1-ol, butan-1-ol, butan-2-ol, propan-1-ol (d) Propan-1-ol, butan-2-ol, butan-1-ol, pentan-1-ol Ans 1. (d) Propan-1-ol, butan-2-ol, butan-1-ol, pentan-1-ol Que 2. Phenol is less acidic than ______________. (a) Methanol (b) p-methylphenol (c) o-nitrophenol (d) o-methoxyphenol Ans 2. (c) o-nitrophenol Que 3. Which of the following is most acidic? (a) Benzyl alcohol (b) Ethanol (c) Phenol (d) m-Chlorophenol Ans 3. (d)…
Case Study Questions Haloalkanes and Haloarenes Class 12 Chemistry Case Study Questions Haloalkanes and Haloarenes Class 12 Chemistry 1. Read the passage given below and answer the following questions: Nucleophilic substitution reactions are of two types; substitution nucleophilic bimolecular (SN2) and substitution nucleophilic unimolecular (SN1) depending on molecules taking part in determining the rate of reaction. The reactivity of alkyl halide towards SN1 and SN2 reactions depends on various factors such as steric hindrance, stability of intermediate or transition state, and polarity of the solvent. SN2 reaction mechanism is favoured mostly by primary alkyl halide or…
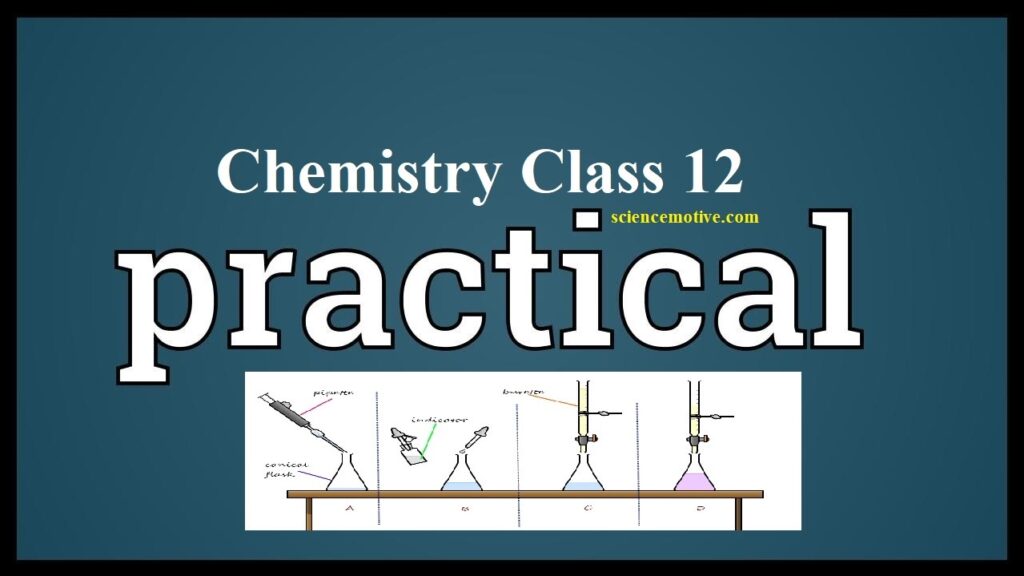
Chemistry Practical Class 12 Chemistry Practical Class 12 TITRATION or VOLUMETRIC ANALYSIS Aim: – To prepare M/50 Oxalic acid solution and determine the molarity, strength of the given potassium permanganate solution. Theory: In this titration oxalic acid acts as a reducing agent and potassium permanganate acts as an oxidising agent. So, the reaction between oxalic acid and potassium permanganate is a redox reaction. In this redox reaction, carbon of oxalic acid gets oxidized to carbon dioxide and manganese present in potassium permanganate, which is in the +7 oxidation state gets reduced to colourless Mn2+ state. Chemistry Practical Class 12 Chemical…
CBSE Class 12 Chemistry Solved Sample Paper 2021-22 (Term-1) CBSE Class 12 Chemistry Solved Sample Paper 2021-22 (Term-1) Subject: Chemistry (043) Time: 90 Minutes Max. Marks: 35Steven Wright, Known Wife Beater, Gets Suspended 80 Games For Doing Steroids – Deadseriousness anadrol 50 prices activity – which anabolic steroid is best for bodybuilding, which anabolic steroids to take – playarcade.fun General Instructions: 1. The Question Paper contains three sections. 2. Section A has…
Assertion Reason Questions on d and f Block Elements Assertion Reason Questions on d and f Block Elements (i) Both assertion and reason are true, and the reason is the correct explanation of the assertion. (ii) Both assertion and reason are true but the reason is not the correct explanation of assertion. (iii) Assertion is not true but the reason is true. (iv) Both assertion and reason are false. Que 1. Assertion: Transition metals show variable valence. Reason: Due to a large energy difference between the ns2 and (n-1)d electrons. Ans 1. (iv) Que 2. Assertion: In transition…
Important MCQs on Biomolecules Important MCQs on Biomolecules Que 1. Which of the following statements is not correct? i) Some antiseptics can be added to soaps. ii) Dilute solutions of some disinfectants can be used as antiseptics. iii) Disinfectants are antimicrobial drugs. iv) Antiseptic medicines can be ingested. Ans 1. (iv) Que 2. Which is the correct statement about birth control pills? i) Contain estrogen only. ii) Contain progesterone only. iii) Contain a mixture of estrogen and progesterone derivatives. iv) Progesterone enhances ovulation. Ans 2. (iii) Que 3. Which statement about aspirin is not true i) Aspirin belongs to…
Biomolecules Class 12 Questions and Answers Biomolecules Class 12 Questions and Answers Que 1. What are monosaccharides? Ans 1. They are single carbohydrates, i.e., polyhydroxy aldehydes (aldoses) or polyhydroxy ketones (ketoses) which cannot be broken into lower sugars upon hydrolysis. They have the general formula, CnH2nOn, where n varies from 3 to 9 carbon atoms. The common monosaccharides are glucose, fructose and ribose, etc. Que 2. What is glycosidic linkage? Give one example Ans 2. The two monosaccharide units are joined together through an oxide (ethereal) linkage formed by the loss of a molecule of H2O. Such a linkage between…
Biomolecules Questions and Answers PDF Biomolecules Questions and Answers PDF Que 1. What are monosaccharides? Ans 1. The simplest carbohydrates that cannot be hydrolyzed to smaller molecules are known as monosaccharides e.g., glucose, fructose, etc. Que 2. What are reducing sugars? Ans 2. The sugars which reduce Tollen’s reagent, Fehling’s Solutions etc. are called reducing sugars. e.g., all monosaccharides, disaccharides except sucrose. Que 3. What is the structural feature characterizing reducing sugar? Ans 3. The reducing sugars have a free aldehydic or ketonic group. Que 4. What are carbohydrates generally optically active? Ans 4. Due to the presence of…
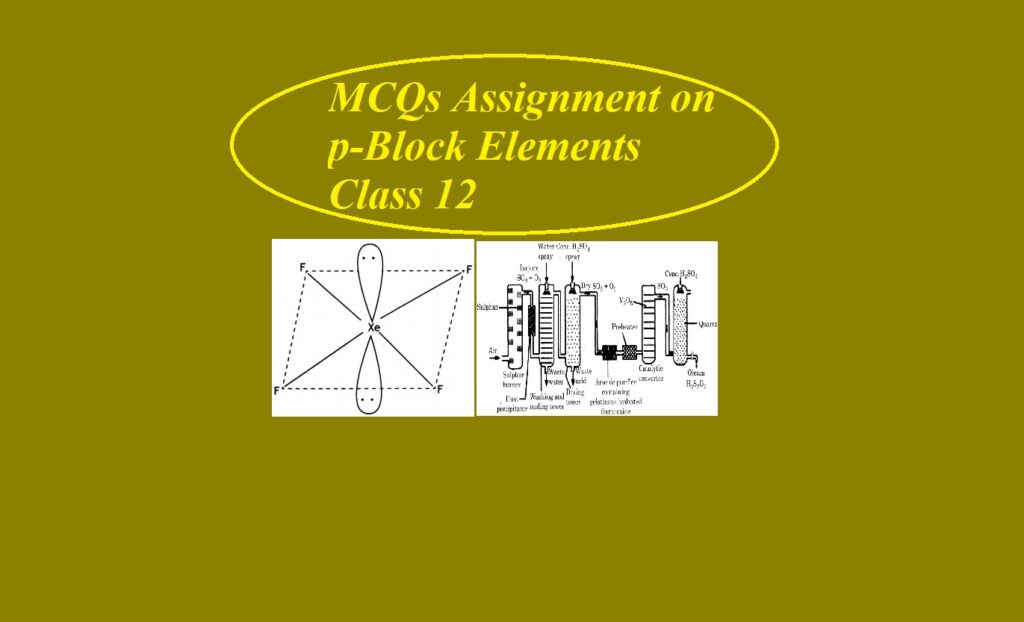
MCQs Assignment on p-Block Elements Class 12 MCQs Assignment on p-Block Elements Class 12 Que 1. Arrange the following in order of increasing acidity H3PO4, H2CO3, HCl, HI (a) H3PO4, HCl, H2CO3, HI (b) H3PO4, H2CO3, HCl, HI (c) H2CO3, H3PO4, HCl, HI (d) None of these Que 2. Which of the following oxides is the most acidic? (a) N2O5 (b) P2O5 (c) As2O5 (d) Sb2O5 Que 3. On heating KClO3, we get (a) KClO2 + O2 (b) KCl + O2 (c) KCl + O3 …