Que. Write down the conclusion made from the α-scattering experiment and the defects of the Rutherford model of the atom.
Ans. Conclusions of α-scattering experiment
(a) Most of the space in the atom is empty as most of the α-particles passed undeflected.
(b) A few positively charged α-particles were deflected. This shows that they are deflected by the small positively charged body in the atom.
(c) The positively charged body called the nucleus is very small of radius 10–15 m as compared to the radius of the atom which is about 10–10 m.
Defects:
(i) Position of electrons: The exact position of the electrons from the nucleus was not mentioned.
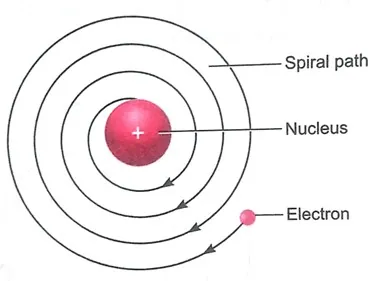
(ii) Stability of atom: According to electromagnetic theory, when a charged body moves under the influence of an attractive force, it loses energy continuously in the form of electromagnetic radiation. The electron should therefore continuously emit radiation and lose energy. As a result of this, a moving electron will come closer to the nucleus through a spiral path, it should ultimately fall into the nucleus. So, Rutherford’s model of atoms could not account for the stability of atoms.
Continuous loss of energy



