Some Basic Concepts of Chemistry Class 11 – MCQs (Answers)
Ans 1.
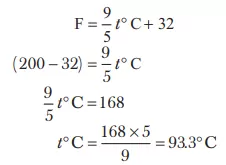
Ans 2. 6.023 × 1023 has four significant figures. The total number of digits in a number including the last digit whose value is uncertain is called the number of significant figures.
Ans 3. All non-zero digits are significant. Hence, there are 6 significant figures in 10.3406 g.
Ans 4. 0.02856 × 298.15 × 0.112 /0.5785
Ans 5. Precision refers to the closeness of various measurements for the same quantity. Accuracy is the agreement of a particular value to the true value of the result. Thus, A and B are precision and accuracy respectively.
Ans 6. As we know, the least count of an instrument is equal to the most possible error of the instrument, hence most possible error of the instrument will be 0.01 cm.
Ans 7. All the given statements are correct about molecular mass.
Ans 8. Molecular mass of urea (NH2CONH2) is
= 2 × (Atomic mass of N) + 4 × (Atomic mass of H) + (Atomic mass of C) + (Atomic mass of O) = 60
Mass percentage of N = (28/60) × 100 = 46.67 %
Ans 9.
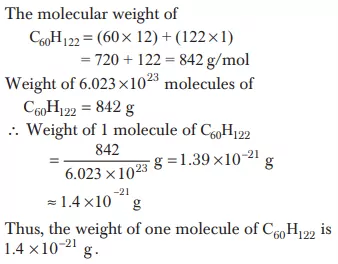
Ans 10. One mole of any substance contains 6.022 × 1023 atoms/ molecules. Hence, the number of millimoles of H2SO4 = Molarity × Volume (in mL) = 0.02 × 100 = 2 × 10−3 mol
Number of molecules = number of moles × NA
= 2 × 10−3 × 6.022 × 1023 = 12.044 × 1020 molecules
Ans 11. In 18 g of water, the number of H2O molecules is NA .
18 mg of water, the number of H2O molecules would be =
![]()
= 10-3 × NA
Ans 12. As 22400 mL water contains water molecules = 6.023 × 1023 molecules
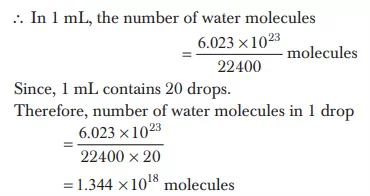
Ans 13.
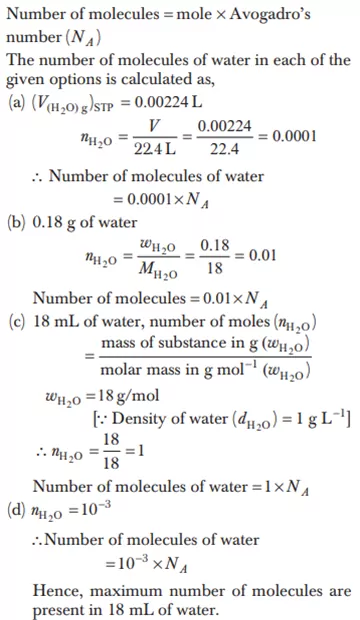
Ans 14.

Ans 15. Molecular mass of CO2 = 1 × 12 + 2 × 16 = 44 g
1 g molecule of CO2 contains 1 g atoms of carbon
؞ 44 g of CO2 contain C = 12 g atoms of carbon
% of C in CO2 = 12/44 × 100 = 27.27 %
Some Basic Concepts of Chemistry Class 11
Ans 16.
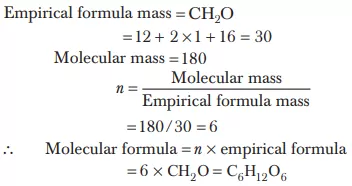 Ans 17.
Ans 17.
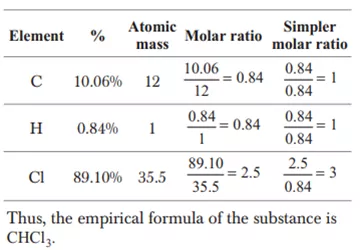
Ans 18. 2Na2HPO4 + NaH2PO4 + 2(NH2)2 CO → Na5P3O10 + 4NH3 + 2CO2
Hence, the stoichiometric ratio of sodium dihydrogen orthophosphate and sodium hydrogen orthophosphate is 2:1
Ans 19. The molar mass of sulphuryl chloride is 32 + 16 + 16 + 35.5 + 35.5 = 135 g/mol
The number of gram molecules in 13.5 g of sulphuryl chloride is = 13.5/135 = 0.1 mol
Ans 20. One litre of the solution
Ans 21. (e) the assertion is false but the reason is true
Ans 22. (e) the assertion is false but the reason is true
Ans 23. (c) the assertion is true but the reason is false.
Ans 24. (e) the assertion is false but the reason is true
Ans 25. (e) the assertion is false but the reason is true
Some Basic Concepts of Chemistry Class 11
Skip to PDF content


