Environmental Chemistry Class 11
Environmental Chemistry class 11 chapter contains all important topics involved. The topics covered are mentioned below
(i) ENVIRONMENTAL CHEMISTRY
(ii) ENVIRONMENTAL POLLUTION
(iii) ATMOSPHERIC POLLUTION
(a) Tropospheric Pollution
(b) Stratospheric Pollution
………………………………………………………………………………………………………………
(i) ENVIRONMENTAL CHEMISTRY
It is the study of chemical processes that occur in water, air, terrestrial and living environments, and the effects of human activity on them.
OR
Environmental Chemistry is the scientific study of the chemical and biochemical phenomena that occur in natural places.
Environmental Chemistry is an interdisciplinary science that includes atmospheric, aquatic, and soil chemistry, as well as heavily relying on analytical chemistry and being related to environmental and other areas of science.
Environmental Chemistry involves first understanding how the uncontaminated environment works, which chemicals in what concentrations are present naturally, and with what effects. Without this, it would be impossible to accurately study the effects humans have on the environment through the release of chemicals.
Environmental Chemistry class 11
(ii) ENVIRONMENTAL POLLUTION:
Environmental pollution is defined as “the contamination of the physical and biological components of the earth/atmosphere system to such an extent that normal environmental processes are adversely affected.
OR
Environmental pollution is the effect of undesirable changes in our surroundings that have harmful effects on plants, animals, and human beings.
Pollutant: A substance, which causes pollution, is known as a pollutant. Pollutants can be solid, liquid or gaseous substances present in greater concentration than in natural abundance and are produced due to human activities or due to natural happenings. Do you know, an average human being requires nearly 12-15 times more air than food? So, even small amounts of pollutants in the air become significant compared to similar levels present in the food.
Pollutants are of two types – degradable and non-degradable.
Degradable pollutants are rapidly broken down by natural processes. E.g. vegetable wastes.
Non-degradable pollutants are not dissociated by natural processes or by micro-organisms and remain in the environment for a long time. E.g. DDT, plastic materials, heavy metals, many chemicals, nuclear wastes etc. once released into the environment are difficult to remove. These pollutants cannot be degraded by natural processes and are harmful to living organisms. In the process of environmental pollution, pollutants originate from a source and get transported by air or water or are dumped into the soil by human beings.
There are different types of pollution: Atmospheric pollution, Water pollution, Air pollution, Solid waste pollution, Noise pollution.
(iii) ATMOSPHERIC POLLUTION:
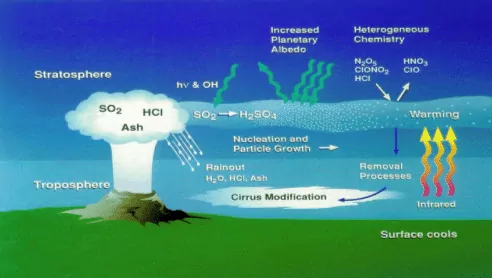
The atmosphere that surrounds the earth is not of the same thickness at all heights. There are concentric layers of air or regions and each layer has a different density. The lowest region of the atmosphere in which human beings along with other organisms live is called Troposphere. It extends up to the height of ~ 10 km from sea level. Above the troposphere, between 10 and 50 km above sea level lies the stratosphere. The troposphere is a turbulent, dusty zone containing air, much water vapour, and clouds. This is the region of strong air movement and cloud formation. The Stratosphere, on the other hand, contains dinitrogen, dioxygen, ozone, and little water vapour. Atmospheric pollution is generally studied as tropospheric and stratospheric pollution.
Atmospheric pollution is the presence of undesirable material in the air in quantities large enough to produce harmful effects.
Environmental Chemistry class 11
(a) Tropospheric Pollution: Tropospheric pollution occurs due to the presence of undesirable solid or gaseous particles in the air. The following are the major gaseous and particulate pollutants present in the troposphere:
1. Gaseous air pollutants: These are oxides of sulphur, nitrogen and carbon, hydrogen sulphide, hydrocarbons, ozone, and other oxidants.
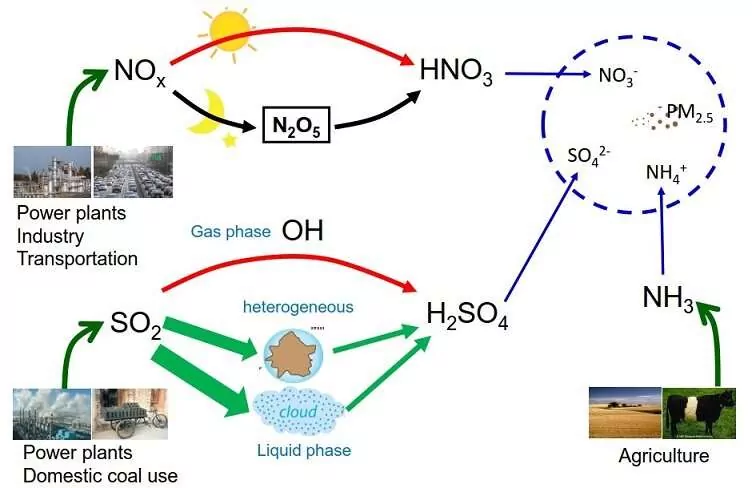
(a) Oxides of Sulphur: Oxides of sulphur are formed when fossil fuel having sulphur is burnt. However, sulphur dioxide is a poisonous gas, which is harmful to both plants and animals. Even low concentration of sulphur dioxide causes respiratory diseases like asthma, bronchitis, emphysema in human beings. Moreover, it causes irritation to the eye. A high concentration of SO2 leads to stiffness of flower buds. Uncatalyzed oxidation of sulphur dioxide is slow. However, in presence of other air pollutants, it facilitates speedy oxidation of sulphur dioxide to trioxide.
2SO2(g) + O2(g) → 2SO3(g).
The reaction can also take place in presence of ozone and hydrogen peroxide.
SO2(g) + O3(g) → SO3(g) + O2(g)
SO2(g) + H2O2(l) → H2SO4(aq)
(b) Oxides of Nitrogen: Dinitrogen and dioxygen are the main constituents of air. These gases don’t react with each other under normal temperatures. However, at high altitudes, when lightning strikes, they combine to form oxides of nitrogen. One of the oxides, NO2 is oxidised to nitrate ion, NO3– which is washed into the soil where it acts as a fertilizer. Also, in an automobile engine, when fossil fuel is burnt, dinitrogen and dioxygen combine to yield quantities of nitric oxide(NO) and nitrogen dioxide(NO2).
N2(g) + O2(g) → 2NO(g)
NO reacts instantly with oxygen to give NO2
2NO(g) + O2(g) → 2NO2(g)
Rate of production of NO2 is faster when nitric oxides react with ozone in the stratosphere.
2NO(g) + O3(g) → 2NO2(g) + O2(g)
The irritant red haze in the traffic and congested places is due to oxides of nitrogen. Higher concentrations of NO2 damage the leaves of plants and retard the rate of photosynthesis. Nitrogen dioxide is a lung irritant that can lead to acute respiratory disease in children. It is toxic to living tissues also. Nitrogen dioxide is also harmful to various textile fibers and metals.
Environmental Chemistry class 11
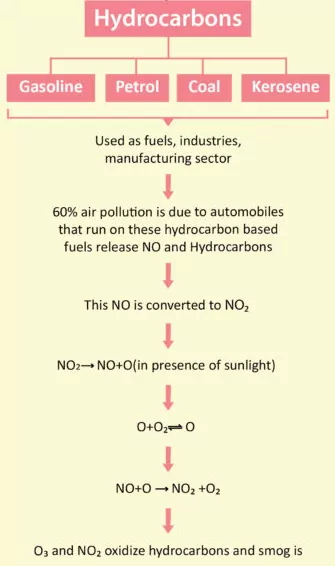
(c) Hydrocarbons: Hydrocarbons are composed of hydrogen and carbon and are formed by incomplete combustion of fuel in automobiles. Also, hydrocarbons are carcinogenic. Moreover, they harm plants by causing aging, breakdown of tissues, and shedding of leaves, flowers, and twigs.
(d) Oxides of Carbon:
1). Carbon Monoxide: It is colorless and odourless gas, however it is highly poisonous in nature. It blocks delivery of oxygen to organs and tissues. Carbon monoxide is generated from incomplete combustion of coal, firewood, petrol, etc. Carbon monoxide when entering in the human body, it combines with haemoglobin to form carboxyhemoglobin. Moreover, carboxyhemoglobin is 300 times stable than the oxygen hemoglobin complex. Thus it prevents the transfer of oxygen to body organs. This also decreases the oxygen-carrying capacity of the blood. It results in headaches, weak eyesight, nervousness, and cardiovascular disorder.
2). Carbon dioxide: It is released into the atmosphere by burning fossil fuels and decomposition of limestone during the manufacture of cement. Carbon dioxide gas forms 0.03% of the atmosphere. With an increase in the use of fossil fuels a large amount of carbon dioxide is released into the atmosphere. Carbon dioxide is used by green plants to prepare their food.
Environmental Chemistry class 11
Global Warming and Greenhouse Effect
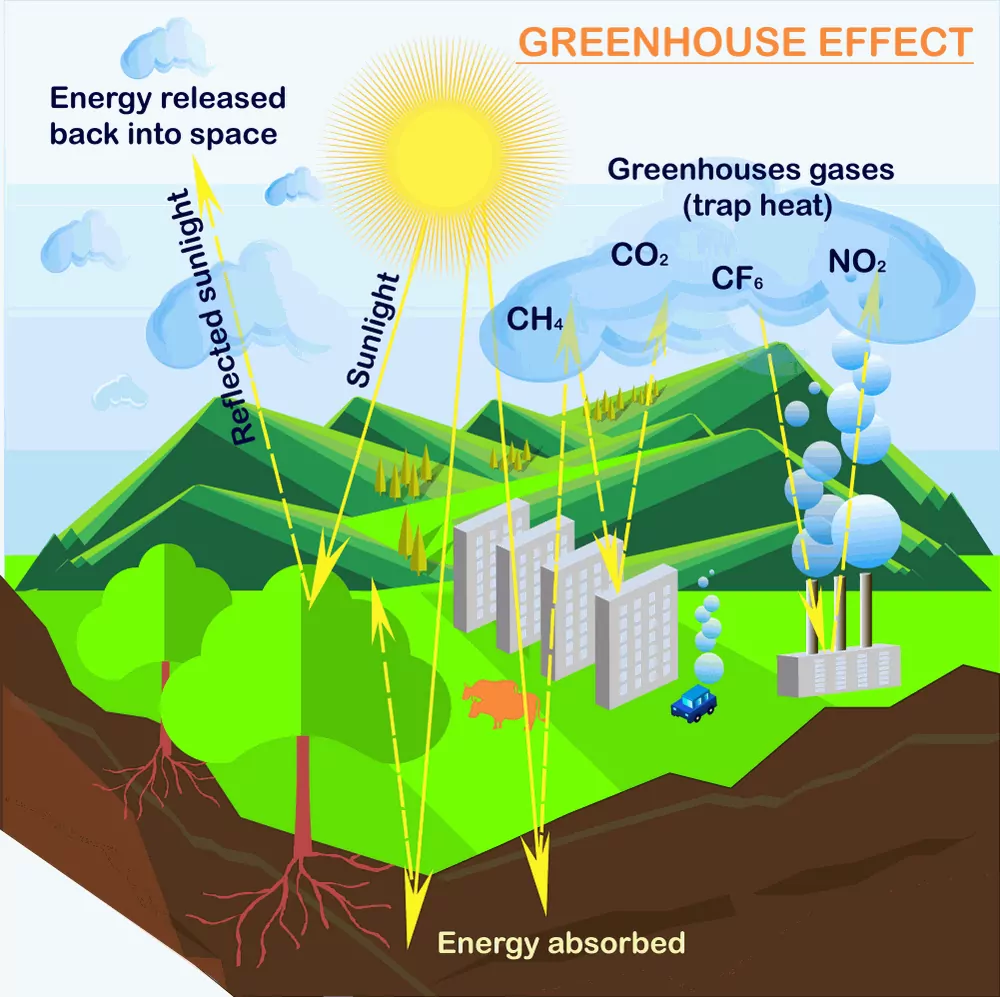
About 75 % of the solar energy reaching the earth is absorbed by the earth’s surface, which increases its temperature. The rest of the heat radiates back to the atmosphere. Some of the heat is trapped by gases such as carbon dioxide, methane, ozone, chlorofluorocarbon compounds (CFCs), and water vapour in the atmosphere. Thus, they add to the heating of the atmosphere. This causes global warming. We all know that in cold places flowers, vegetables and fruits are grown in glass-covered areas called greenhouse. In a greenhouse, solar radiations pass through the transparent glass and heat up the soil and the plants. The warm soil and plants emit infrared radiations. Since glass is opaque to infrared radiations (thermal region), it partly reflects and partly absorbs these radiations. This mechanism keeps the energy of the sun trapped in the greenhouse. Similarly, carbon dioxide molecules also trap heat as they are transparent to sunlight but not to heat radiation. If the amount of carbon dioxide crosses the delicate proportion of 0.03 percent, the natural greenhouse balance may get disturbed. Carbon dioxide is a major contributor to global warming. Besides carbon dioxide, other greenhouse gases are methane, water vapour, nitrous oxide, CFCs, and ozone.
Environmental Chemistry class 11
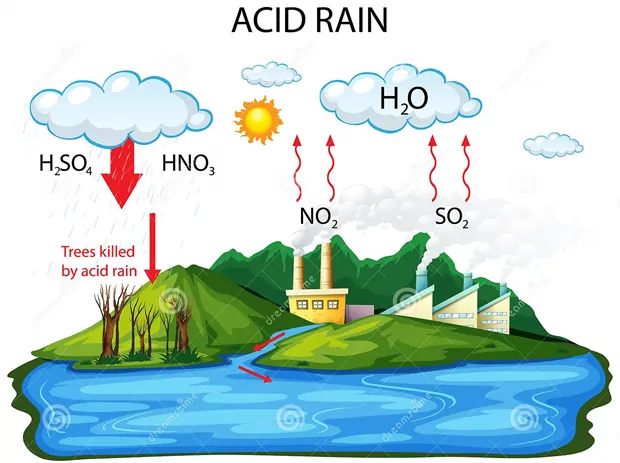
Acid rain
We are aware that normally rainwater has a pH of 5.6 due to the presence of H+ ions formed by the reaction of rainwater with carbon dioxide present in the atmosphere.
H2O (l) + CO2 (g) → H2CO3 (aq)
H2CO3 (aq) → H+ (aq) + HCO3– (aq)
When the pH of the rain water drops below 5.6, it is called acid rain. Acid rain refers to the ways in which acid from the atmosphere is deposited on the earth’s surface. Oxides of nitrogen and sulphur which are acidic in nature can be blown by wind along with solid particles in the atmosphere and finally settle down either on the ground as dry deposition or in water, fog and snow as wet deposition.
Environmental Chemistry class 11
2. Particulate pollutants: Particulates pollutants are the minute solid particles or liquid droplets in the air. These are present in vehicle emissions, smoke particles from fires, dust particles, and ash from industries. Particulates in the atmosphere may be viable or non-viable. The viable particulates e.g., bacteria, fungi, moulds, algae, etc., are minute living organisms that are dispersed in the atmosphere. Human beings are allergic to some of the fungi found in the air. They can also cause plant diseases. Non-viable particulates may be classified according to their nature and size as follows:
Smoke particulate: It contains solid, or, a mixture of solid and liquid particles formed during combustion of organic matter. Some examples are cigarette smoke, smoke from the burning of fossil fuels, garbage, dry leaves, oil smoke, etc.

Dust particulate: Dust is made of fine solid particles, over 1µm. It is mainly produced during the crushing, grinding, and attribution of solid materials. Some examples of particulate emissions are sand, sawdust, pulverized coal, cement, fly ash from factories, etc.
 Environmental Chemistry class 11
Environmental Chemistry class 11
Mist particulate: It is produced by particles of spray liquids and by condensation of vapors in air. Some examples of this type are sulphuric acid mist and herbicides and insecticides that miss the target and travel through air and forms mists.

Fumes particulate: Fumes are formed by condensation of vapors during sublimation, distillation, boiling, and several other chemical reactions. Organic solvents, metals, and metallic oxides mostly form fume particles.

The effect of particulate pollutants are largely dependent on the particle size. Airborne particles such as dust, fumes, mist, etc., are dangerous for human health. Particulate pollutants bigger than 5 microns are likely to lodge in the nasal passage, whereas particles of about 10 microns enter into the lungs easily.
Environmental Chemistry class 11
Smog
The word smog is derived from smoke and fog. This is the most common example of air pollution that occurs in many cities throughout the world. There are two types of smog:
(a) Classical smog occurs in a cool humid climate. It is a mixture of smoke, fog, and sulphur dioxide. Chemically it is a reducing mixture and so it is also called reducing smog.

(b) Photochemical smog occurs in a warm, dry, and sunny climate. The main components of the photochemical smog result from the action of sunlight on unsaturated hydrocarbons and nitrogen oxides produced by automobiles and factories. Photochemical smog has a high concentration of oxidising agents and is, therefore, called as oxidizing smog.
Environmental Chemistry class 11
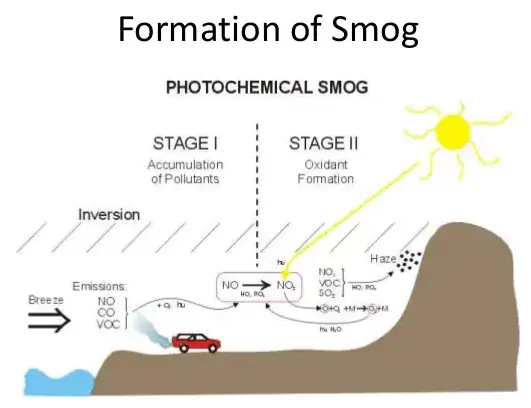
Formation of photochemical smog: When fossil fuels are burnt, a variety of pollutants are emitted into the earth’s troposphere. Two of the pollutants that are emitted are hydrocarbons (unburnt fuels) and nitric oxide (NO). When these pollutants build up to sufficiently high levels, a chain reaction occurs from their interaction with sunlight in which NO is converted into nitrogen dioxide (NO2). This NO2 in turn absorbs energy from sunlight and breaks up into nitric oxide and a free oxygen atoms (Fig. 14.2).
NO2(g) → NO(g) + O(g) ……………..(i)
Oxygen atoms are very reactive and combine with the O2 in air to produce ozone.
O(g) + O2 (g) → O3 (g) ……………………..(ii)
The ozone formed in the above reaction (ii) reacts rapidly with the NO(g) formed in the reaction (i) to regenerate NO2. NO2 is a brown gas and at sufficiently high levels can contribute to haze. NO (g) + O3 (g) → NO2 (g) + O2 (g) ……….(iii)
Ozone is a toxic gas and both NO2 and O3 are strong oxidising agents and can react with the unburnt hydrocarbons in the polluted air to produce chemicals such as formaldehyde, acrolein, and peroxyacetyl nitrate (PAN).
3CH4 + 2O3 → 3CH2= O + 3H2O
Formaldehyde
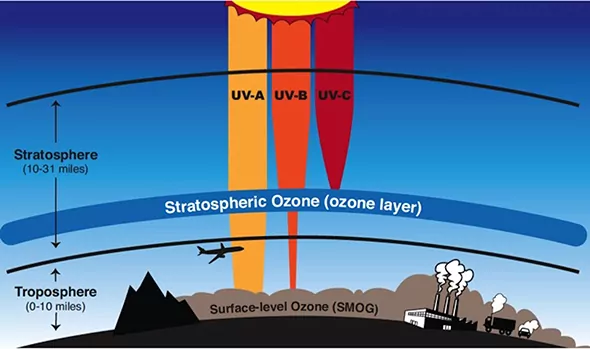
Stratospheric Pollution: Stratosphere is the layer of the earth’s atmosphere just above the troposphere and below the mesosphere. The upper stratosphere consists of a considerable amount of ozone (O3), which protects us from the harmful ultraviolet (UV) radiations coming from the sun. These radiations cause skin cancer in humans. Therefore, it is important to maintain.
 Environmental Chemistry class 11
Environmental Chemistry class 11
Formation and Breakdown of Ozone: The upper stratosphere consists of a considerable amount of ozone (O3), which protects us from the harmful ultraviolet (UV) radiations (λ = 255 nm) coming from the sun.
In the stratosphere (10-15 km from the earth’s surface), the O2 is partially converted into O3. It is a two-step process.
Step 1. The UV radiation coming from the sun has sufficient energy to split the O2 into 2 oxygen radicals.
O2 + UV → 2O
Step 2. The oxygen radicals react with more oxygen molecules to form ozone.
O2 + O + UV → O3
Ozone absorbs U.V radiation and again breaks into oxygen molecules and oxygen radicals.
O3 + UV → O2 + O
Heat is given out in this process which warms up the stratosphere. For this reason, the stratosphere is a zone of increasing temperature.
O2 + UV → O2 + O + x kcal
In this way, O3 cycle is completed in this region.
Depletion of the ozone layer in the stratosphere.
Due to human activity two compounds NO and CFC, are responsible for the depletion of the ozone layer.
(a) NO Nitric Oxide
NO + O3 → NO2 + O2
NO2 + O → NO + O2
The NO reacts with O3 causing a decrease in the amount of O3 and forms NO2 which reacts with the amount of O3 and forms NO2 which reacts with oxygen radicals for regeneration of NO. Thus there is no net decrease in the amount of NO while amount of O3 decreases.
(b) Chlorofluorocarbons (CFC) or freons:
The freons decompose in the presence of U.V. radiation coming from the sun.
CF2Cl2 + hv → CF2Cl + Cl
CFCl3 + hv → Cl + CFCl2
The reactive chlorine atoms then destroy the ozone layer through the following sequence of reactions which are repeated because chlorine atoms are regenerated in the second reaction.
Cl + O3 → ClO + O2
ClO + O → Cl + O2
One molecule of CFC can destroy more than one thousand O3 molecules in the stratosphere.
Environmental Chemistry class 11