Nucleophilic Substitution and Mechanism of Haloalkanes
The Post Nucleophilic Substitution and Mechanism of Haloalkanes is all about various nucleophilic substitution reactions of haloalkanes along with the followed mechanisms.
- Chemical Reactions of Haloalkanes:
- Nucleophilic Substitution Reactions: Due to the presence of a positive charge on the carbon atom it can be easily attacked by nucleophiles to give nucleophilic substitution reactions as well as elimination and reduction reactions.
1). Reaction with aqueous alkali: Haloalkanes react with aq. NaOH or KOH to form alcohols.
R-X + KOH(aq) → R-OH + KX
CH3-CH2-Br + KOH(aq) → CH3-CH2-OH + KBr
2). Reaction with water: Haloalkanes react with water to form alcohols.
R-X + H2O → R-OH + HX
3). Reaction with Sodium alkoxide (R-ONa) [Williamson’s ether synthesis]
Haloalkanes react with sodium alkoxide to give ethers (R-O-R).
R-X + R-ONa → R-O-R + KX
CH3-Br + CH3-CH2-ONa → CH3-O-CH2-CH3 + NaBr
4). Reaction with Sodium iodide (NaI) [Finkelstein Reaction]: Alkyl halides
(Chlorides or Bromides) react with sodium alkoxide to form alkyl iodides.
R-X + NaI → R-I + NaX [X = Cl or Br]
5). Reaction with ammonia [Hoffmann’s reaction]: Alkyl halides react with alcoholic ammonia to give a mixture of 10, 20 and 30 amines and quaternary ammonium salt.
R-X + NH3 → R-NH2 + R2NH + R3N + R4N+X–
If ammonia is in excess, only primary amine is formed.
6). Reaction with KCN: Alkyl halides react with alcoholic KCN to give alkane nitriles or alkyl cyanides (R-CN).
7). Reaction with Silver cyanide (AgCN): Alkyl halides react with AgCN to give alkyl isocyanides or carbyl amines (R-NC).
R-X + AgCN → R-NC + AgX
CN– is an ambident nucleophile. i.e. here both C and N contain lone pair of electrons and can bind to the carbon atom of the alkyl group either through C or through N. Another e.g. is NO2–
Reaction with KCN gives alkyl cyanides. This is because KCN is mainly ionic and gives CN– ions in solution. So, both C and N are free to donate electron pairs. But the C – C bond is stronger than the C – N bond. So, cyanides are formed as the major product. But AgCN is mainly covalent and the only N is free to donate an electron pair. So, isocyanides are the main product.
8). Reaction with Silver nitrite (AgNO2): Alkyl halides react with AgNO2 to give nitroalkane (R-NO2)
R-X + AgNO2 → R-NO2 + AgX
9). Reduction: Alkyl halides when reduced with lithium aluminium hydride (LiAlH4) to give alkane.
R-X + [H] → R-H + HX
- Mechanism of Nucleophilic Substitution Reactions: There are two types of mechanisms:
- Bimolecular Nucleophilic Substitution (SN2)
- Unimolecular Nucleophilic Substitution (SN1)
Bimolecular Nucleophilic Substitution (SN2):
Characteristics of SN2:
i). The rate of the reaction depends on the concentration of alkyl halide and on the concentration of the nucleophile.
CH3 -CI + OH – → CH3 -OH + CI
Thus Rate = K [CH3-CI][OH–]
In this reaction, two species CH3-CI and OH– are involved in the rate-determining step hence reaction is bimolecular reaction.
ii). It takes place in one step. They are Concerted Reactions i.e. bond breaking and bond making takes place in one step. No intermediate is formed.
iii). Product formation takes place by formation of transition state 
It takes place in one step. They are Concerted Reactions. bond breaking and bond making takes place in one step. No intermediate is formed.
Stereochemistry of SN2: The attack of nucleophile occurs from a direction opposite to the one from where the halogen atom leaves. Thus, the inversion of the configuration takes place.

Bulky groups cause steric hindrance in the reaction and thus reactivity of alkyl halides is,
Methyl halide > 1° > 2° > 3°
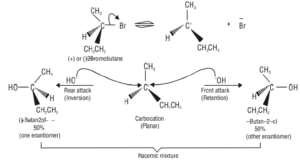
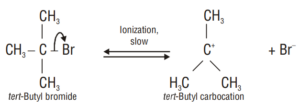
Unimolecular Nucleophilic Substitution (SN1):
Step 1. This step is slow and reversible so it is the rate-determining step of the reaction.
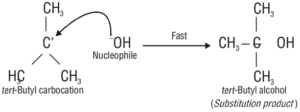
Step 2. In the second step, the carbocation being a reactive chemical species is immediately attacked by the nucleophile i.e., OH¯ ion to give the substitution product, i.e., tertbutyl alcohol. This step is fast and hence does not affect the rate of the reaction.
Stereochemistry of SN1 reactions:
In SN1 reactions, if the alkyl halide is optically active, then the product is a racemic mixture, i.e., SN1 reactions are accompanied by racemization. This is due to the reason that carbocations are the intermediates in SN1 reactions. Since carbocations being sp2 hybridized are planar (achiral) species, therefore, the attack of the nucleophile on it can occur from both the faces.