Electrochemical Cells Nernst Equation and Gibbs Energy
Syllabus:-
- Electrolytic cells & Galvanic cells
- The function of Salt Bridge
- Redox reaction, EMF of the cell, standard electrode potential
- Standard hydrogen electrode (SHE)
- Nernst equation and its application to Chemical cell
- Equilibrium Constant from Nernst Equation
- Electrochemical Cell and Gibbs Energy
Electrochemistry: It is a branch of chemistry that deals with the relationship between chemical energy and electrical energy and their interconversions.
Redox Reactions: Oxidation is the process that involves the loss of electrons & reduction is a process in which it involves the gain of electrons. The reactions which involve both that reaction simultaneously are called a redox reaction.
Electrochemical Cells: These are devices that convert the chemical energy of some redox reactions to electrical energy. They are also called Galvanic cells or Voltaic cells. An example for a Galvanic cell is Daniel cell.
It is constructed by dipping a Zn rod in ZnSO4 solution and a Cu rod in CuSO4 solution. The two solutions are connected externally by a metallic wire through a voltmeter and a switch and internally by a salt bridge.
A salt bridge is a U-tube containing an inert electrolyte like NaNO3 or KNO3 in a gelly like substance. The functions of a salt bridge are:
- To complete the electrical circuit
- To maintain the electrical neutrality in the two half cells.
- Cu2+ + 2 e– → Cu(s) (reduction half reaction)
- Zn(s) → Zn2+ + 2 e– (oxidation half reaction)
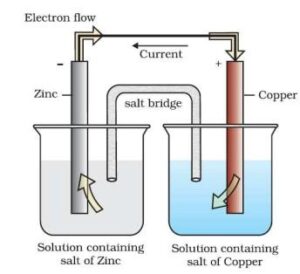
These reactions occur in two different portions of the Daniel cell. The reduction half-reaction occurs on the copper electrode while the oxidation half-reaction occurs on the zinc electrode. These two portions of the cell are also called half-cells or redox couples. The copper electrode may be called the reduction half-cell and the zinc electrode, the oxidation half-cell.
Electrochemical Cells Nernst Equation and Gibbs Energy
Electrode Potential: This tendency of a metal to lose or gain an electron when it is in contact with its own solution is called electrode potential.
Standard electrode potential (E0): The electrode potential measured at standard conditions.ie at 298K,1 atm pressure, and at 1 molar concentration.
Standard hydrogen electrode (SHE): The reference electrode used to measure single electrode potential. Its potential is assumed to be zero. It consists of a platinum wire dipped in HCl of 1 molar concentration. Hydrogen gas at 1 atm. is passed through the solution. The electrode can be represented as Pt, H2 /H+(1M).
Nernst Equation For A Cell Reaction:-
Let us consider a general equation
aA + bB → cC + dD
E = E0cell – 2.303 RT × log [C]c [D]d
nF [A]a [B]b
Where E0 is the standard electrode potential,
R is the gas constant (8.314 JK–1 mol–1)
F is Faraday constant (96500 C mol–1)
T is the temperature in Kelvin.
Nernst equation can be written as:
Putting the above values Nernst equations can be written as
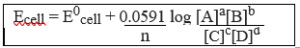
Equilibrium Constant from Nernst Equation:
 Electrochemical Cells Nernst Equation and Gibbs Energy
Electrochemical Cells Nernst Equation and Gibbs Energy
Electrochemical Cell and Gibbs Energy of the Reaction:
Electrical work done in one second is equal to electrical potential multiplied by total charge passed. The reversible work done by a galvanic cell is equal to a decrease in its Gibbs energy and therefore if the emf of the cell is E and nF is the amount of charge passed and ∆G is the Gibbs energy of the reaction, then
∆G = – nFEcell
If the concentration of all the reacting species is unity, then Ecell = E0cell and we have
∆G0 = – nFE0 cell
Thus, from the measurement of E0cell, we can calculate the standard Gibbs energy of the reaction.



