Acids Bases and Salts Class 10 Notes
ACIDS
Acid is a substance that furnishes H+ ions or H3O+ ions when dissolved in water. Acids have one or more replaceable hydrogen atoms. The word acid is derived from the Latin name ‘Acidus’ which means Sour Taste. Substances with a ‘sour taste’ are acids. Lemon juice, vinegar and grape juice have a sour taste, so they are acidic.
Substances that turn blue litmus solution red are called acids.
CLASSIFICATION OF ACIDS:
1. Based on Source: Acids are of two types Mineral acids (Inorganic acid), Organic acid.
(a) Acids that are obtained from minerals and rocks are called Mineral acids. For e.g., H2SO4 (Sulphuric acid), HNO3 (Nitric acid) and HCl (Hydrochloric acid).
(b) Acids that are extracted from plants and animals are called Organic acids. For e.g. Citric acid (C6H8O7), Ascorbic acid (C6H8O6), Tartaric acid (C4H6O6), Lactic acid (C3H6O3), Acetic acid (C2H4O2).
2. Based on ionisation: Acids are classified into two types based on ionisation.
(a) Strong acids: Completely dissociate into its ions in aqueous solutions. Example: Nitric acid (HNO3), Sulphuric acid (H2SO4), Hydrochloric acid (HCl).
(b) Weak acids: Which do not completely dissociate into their ions in aqueous solutions. For example Carbonic acid (H2CO3), acetic acid (CH3COOH).
3. Based on concentration: Depending on the percentage or amount of acid dissolved in water acids are classified into dilute acid and concentrated acid.
(a) Dilute acids: Have a low concentration of acids in aqueous solutions.
(b) Concentrated acids: Have a high concentration of acids in aqueous solutions.
4. Based on their number of Hydrogen ion acids are:
(a) Monoprotic acid: Such type of acid produces one mole of H+ ions per mole of acid, e.g., HCl, HNO3.
(b) Diprotic acid: They can produce two moles of H+ ions per mole of acid, e.g., H2SO4.
(c) Triprotic acid: They produce three moles of H+ ions per mole of acid, e.g., H3PO4.
(d) Polyprotic: They can produce more than three H+ ions per mole of acid.
Acids Bases and Salts Class 10 Notes
CHEMICAL PROPERTIES OF ACIDS:
(a) The reaction of Acids With Metal:
Acids react with active metals to give Salt and Hydrogen gas.
Metal + Acid → Salt + Hydrogen
For Example:
Hydrogen gas and Zinc Chloride are formed when Zinc Metal reacts with Sulphuric acid.
Zn + H2SO4 → ZnSO4 + H2
Hydrogen gas and Sodium Chloride are formed when Sodium Metal reacts with Hydrochloric acid.
2 Na + 2 HCl → 2 NaCl + H2
(b) Reaction of Acids with Metal Carbonate:
Acids react with metal carbonates to form salt and water with the evolution of carbon dioxide gas.
Metal Carbonate + Acid → Salt + Carbon dioxide + Water
For Example:
Sulphuric acid gives calcium sulphate, carbon dioxide gas, calcium sulphate and water when it reacts with calcium carbonate.
CaCO3 + H2SO4 → CaSO4 + CO2 + H2O
Hydrochloric acid reacts with sodium carbonate to form sodium chloride and water with the release of carbon dioxide gas.
Na2CO3(s) + 2 HCl(aq) → 2NaCl(aq) + CO2(g) + H2O(l)
Hydrochloric acid gives carbon dioxide, magnesium chloride and water when it reacts with magnesium carbonate.
MgCO3 + 2HCl → MgCl2 + CO2 + H2O
Hydrochloric acid gives carbon dioxide gas, calcium chloride and water when it reacts with calcium carbonate.
CaCO3 + 2HCl → CaCl2 + CO2 + H2O
(c) Reaction of Acid With Hydrogen Carbonates (Bicarbonates):
Acids give carbon dioxide gas, respective salt and water when they react with metal hydrogen carbonate.
Acid + Metal hydrogen carbonate → Salt + Carbon dioxide + Water
Sodium bicarbonate also reacts with hydrochloric acid to form sodium chloride and water with the release of carbon dioxide gas.
NaHCO3(s) + HCl (aq) → NaCl (aq) + CO2(g) + H2O(l)
Sulphuric acid gives sodium sulphate, carbon dioxide gas and water when it reacts with sodium bicarbonate.
2 NaHCO3 + H2SO4 → Na2SO4 + 2CO2 + 2H2O
Acids Bases and Salts Class 10 Notes
USES of ACIDS:
(i) Sulphuric acid (King of chemicals) is used in the car battery and in the preparation of many other compounds
(ii) Nitric acid is used in the production of ammonium nitrate which is used as fertilizer in agriculture.
(iii) Hydrochloric acid is used as a cleansing agent in the toilet.
(iv) Tartaric acid is a constituent of baking powder.
(v) Salt of benzoic acid (sodium benzoate) is used in food preservation.
(vi) Carbonic acid is used in aerated drinks.
BASES
Substances that change red litmus solution to blue are called bases. They are bitter in taste.
They release hydroxide ions (OH–) when dissolved in water. They have a soapy touch (e.g. Washing soda, caustic soda, and caustic potash).
CLASSIFICATION OF BASES:
1. Based on Ionisation:
(a) Strong Bases: These are bases that ionise completely in an aqueous solution eg. NaOH, KOH.
(b) Weak Bases: These are bases that ionise partially in an aqueous solution eg. NH4OH, Ca(OH)2.
2. Based on their Acidity:
(a) Monoacidic Base: It is a base that ionises in water to give one hydroxide ion per molecule eg. NaOH, KOH.
(b) Diacidic Base: It is a base that ionises in water to give two hydroxide ions per molecule eg. Ca(OH)2, Mg(OH)2.
(c) Triacidic Base: It is a base that ionises in water to give three hydroxide ions per molecule eg. Al(OH)3, Fe(OH)3.
3. Based on the Concentration:
Depending upon the amount of water added to a base, bases are classified as concentrated and dilute Bases.
(a) Concentrated alkali: It is an alkali having a relatively high percentage of alkali in its aqueous solution.
(b) Dilute alkali: It is an alkali having a relatively low percentage of alkali in its aqueous solution.
Acids Bases and Salts Class 10 Notes
CHEMICAL PROPERTIES of BASE:
(a) Reaction of Base with Metals:
When alkali (base) reacts with metal, it produces salt and hydrogen gas.
Alkali + Metal → Salt + Hydrogen
Example 1. Sodium aluminate and hydrogen gas are formed when sodium hydroxide reacts with aluminium metal.
2 NaOH + 2 Al + 2 H2O → 2 NaAlO2 + 2 H2
Example 2. Sodium hydroxide gives hydrogen gas and sodium zincate when reacts with zinc metal.
2 NaOH + Zn → Na2ZnO2 + H2
(b) Reaction of Non-Metallic Oxides with Base:
Non-metallic oxides react with a base to produce Salt and Water
Non-metallic oxide + Base → Salt + Water
Example. Ca(OH)2 + CO2 → CaCO3 + H2O
Non Metallic oxides are said to be acidic in nature because on reacting with a base they produce Salt and Water.
(c) Action of Alkalis/Base with Ammonium Salts:
Alkalis combine with ammonium salts to liberate ammonia.
Alkali + Ammonium salt → Salt + Water + Ammonia
Example. Calcium Hydroxide when reacting with ammonium chloride produces calcium chloride water and ammonia
Ca(OH)2 + NH4Cl → CaCl2 + H2O + NH3
Acids Bases and Salts Class 10 Notes
USES OF BASES:
(i) Magnesium hydroxide is used as a medicine for stomach troubles.
(ii) Ammonium hydroxide is used to remove grease stains from clothes.
(iii) Sodium hydroxide is used in the manufacture of soap.
(iv) Calcium hydroxide is used in whitewashing buildings.
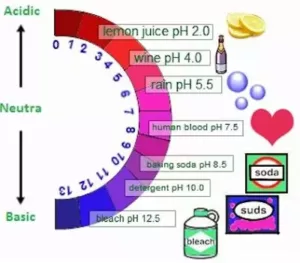
STRENGTH OF ACID OR BASE SOLUTIONS
A scale for measuring hydrogen ion concentration in a solution, called a pH scale has been developed. The p in pH stands for ‘potenz’ in German, meaning power.
p = potential or Power H = Hydrogen
(i) pH of a solution: pH of a solution is the negative logarithm to the base 10 of the hydrogen ion concentration expressed in mole per litre. pH = –log10 (H+).
| pH = 7 | Neutral Solution | H3O+ = OH – |
| pH > 7 | Basic Solution | H3O+ < OH – |
| pH< 7 | Acidic Solution | H3O+ > OH – |
| The range of pH is from 1 to 14 | ||
(ii) Detecting an Acid or a Base: We use litmus solution, phenolphthalein or methyl orange for this purpose. The colour change that they show in acidic and basic solutions are:
| Neutral | Acid | Base | |
| Litmus solution | Purple | Red | Blue |
| Phenolphthalein | Colourless | No change | Pink |
| Methyl orange | Orange | Red | Yellow |
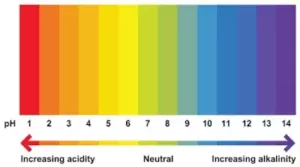
We can also measure the strength of an acid or a base using a pH scale, which looks something like this:
 (iii) pH Sensitivity of Plants & Animals:
(iii) pH Sensitivity of Plants & Animals:
- The human body works in a narrow range of pH 7 to 7.8. Acidity can be lethal for plants and animals.
- the pH of the Digestive System: The stomach secretes HCl to kill bacteria in the food. The inner lining of the stomach protects vital cells from this acidic pH.
- pH and Tooth Decay: Lower pH because sour food and sweet food can cause tooth decay. The pH of the mouth should always be more than 5.5.
- pH as self Defence Mechanism in Plants & Animals: Certain animals like bees and plants like nettle secrete highly acidic substances for self-defence.
- Acid Rain: If the pH of rain is lower than 5.6 we term it ‘acid rain’. This water, when flows in water bodies, makes the water acidic and difficult for aquatic organisms to survive. It also causes harm to monuments like the Taj Mahal.
Acids Bases and Salts Class 10 Notes
INDICATOR
Substances which show the acidic or basic behaviour of other substances by a change in colour are known as the indicator.
(i) Type of Indicator: There are many types of indicators. Some common types of indicators are
Natural Indicator, Olfactory Indicator, Synthetic Indicator, Universal Indicator
(a) Natural Indicator: Indicators obtained from natural sources are called natural indicators. Litmus, turmeric, red cabbage, China rose, etc. are some common natural indicators used widely to show the acidic or basic character of substances. For Eg.
- Litmus – Litmus is obtained from lichens. The solution of litmus is purple in colour. Litmus paper comes in two colours – blue and red.
An acid turns blue litmus paper red.
A base turns red litmus paper blue.
- Turmeric: Turmeric is another natural indicator. Turmeric is yellow in colour. Turmeric solution or paper turns reddish brown with base. Turmeric does not change colour with acid.

- Red Cabbage: The juice of red cabbage is originally purple in colour. The juice of red cabbage turns reddish with acid and turns greenish with the base.
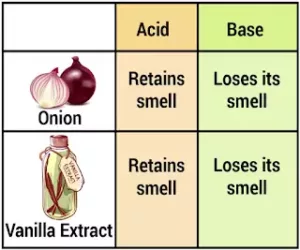
(b) Olfactory Indicator: Substances which change their smell when mixed with acid or base are known as olfactory indicators. For example, onion, vanilla, clove, etc.
- Onion: Paste or juice of onion loses its smell when added to the base. It does not change its smell with acid.
- Vanilla: The smell of vanilla vanishes with the base, but its smell does not vanish with acid. Olfactory indicators are used to ensure the participation of visually impaired students in the laboratory.
(c) Synthetic Indicator: Indicators that are synthesized in the laboratory are known as synthetic indicators. For example; phenolphthalein, methyl orange, etc.
- Phenolphthalein: It is a colourless liquid. It remains colourless with acid but turns pink with a base.
- Methyl orange: It is originally orange in colour. It turns red with acid and turns yellow with the base.
(d) Universal Indicator: In the case of a colourless liquid, an accurate pH can be obtained by adding a universal indicator. It is a mixture of several indicators and shows different colours at different concentrations of hydrogen ions in a solution. For Example:
- A universal indicator produces green colour in a neutral solution, pH = 7.
- The colour changes from blue to violet as pH increases from 7 to 14.
- The colour changes from yellow to pink and then to red as pH decreases from 7 to 1.
Acids Bases and Salts Class 10 Notes
SALTS
A Salt is an ionic compound that is made up of two groups of oppositely charged ions. The ion with a positive charge is called a cation, and the one with a negative charge is called an anion.
Classification of Salts
(1) Normal salts: A normal salt is obtained by the complete neutralization of an acid by a base.
NaOH + HCl → NaCl + H2O
(2) Acid salts: Acid salts are derived by the partial replacement of hydrogen ions of an acid by a metal. When a calculated amount of a base is added to a polybasic acid, acid salt is obtained, as follows.
NaOH + H2SO4 → NaHSO4 + H2O
(3) Basic salts: Basic salts are formed by the partial replacement of hydroxide ions of a diacidic or triacidic base by an acid radical. A basic salt may further react with an acid to give a normal salt.
Pb(OH)2 + HCl → Pb(OH)Cl + H2O
Diacidic base Basic salt
| Family of Salts: Salts can be classified into the following types: Normal or Neutral Salts – A salt that is formed by the complete replacement of the replaceable hydrogen ions of an acid by a metal ion or ammonium ion is called a normal salt. Examples: Neutral Salts: Strong Acid + Strong base pH value is 7. Examples: NaCl, CaSO4 Acidic Salts: Strong Acid + weak base pH value is less than 7. Examples: NH4Cl, NH4NO3 Basic Salts: Strong base + Weak acid pH value is more than 7. Examples: Na2CO3, CH3COONa. |
Acids Bases and Salts Class 10 Notes
Neutralization
The reaction between an acid and a base to form salt and water is called a neutralization reaction.
Acid + Base → Salt + Water
(i) Hydrochloric acid reacts with sodium hydroxide to form sodium chloride and water.
HCl + NaOH → NaCl + H2O
(ii) Sodium sulphate and water are formed when sulphuric acid reacts with sodium hydroxide (a base).
H2SO4 + 2NaOH → Na2SO4 + 2H2O
(iii) Calcium chloride is formed along with water when hydrochloric acid reacts with calcium hydroxide (a base).
2HCl + Ca(OH)2 → CaCl2 + 2H2O
Reaction Of Acid With Metal Oxides
Metal oxides are basic in nature. Thus, when an acid reacts with a metal oxide both neutralize each other. In this reaction, respective salt and water are formed.
Acid + Metal Oxide → Salt + Water
(i) Sulphuric acid reacts with zinc oxide, zinc sulphate, and water are formed.
H2SO4 + ZnO → ZnCl2 + H2O
(ii) Copper oxide (II), a black metal oxide reacts with dilute hydrochloric acid to form a blue-green coloured copper chloride (II) solution.
CuO + 2HCl → CuCl2(aq) + H2O
Acids Bases and Salts Class 10 Notes
Common Salt
(i) Chemical name: Sodium Chloride (NaCl)
(ii) Common salt is a neutral salt and can be prepared in the laboratory by the reaction of sodium hydroxide and hydrochloric acid.
NaOH(aq) + HCl(aq) → NaCl(aq) + H2O(aq)
(iii) It is an important raw material for products of daily use such as NaOH, baking soda, washing soda, and bleaching powder.
Sodium Hydroxide
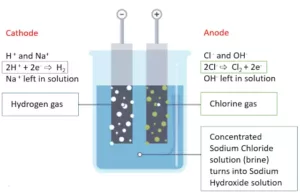
(i) Preparation: It is obtained by the electrolytic decomposition of a solution of sodium chloride (brine). In the process of electrolytic decomposition of brine (Brine: aqueous solution of sodium chloride), brine decomposes to form sodium hydroxide. In this process, chlorine is obtained at the anode and hydrogen gas is obtained at the cathode as by-products. This whole process is known as the Chlor-Alkali process (i.e. ‘Chlor’ for Chlorine and ‘Alkali’ for Sodium Hydroxide).
2NaCl + 2H2O → 2NaOH + Cl2 + H2
Washing Soda
(i) Chemical formula: Na2CO3.10H2O
(ii) Sodium hydrogen carbonate, on heating decomposes to give sodium carbonate with the release of Carbon dioxide gas. Re-crystallisation of sodium carbonate produces washing soda.
2NaHCO3 → Na2CO3 + H2O + CO2
Na2CO3 + 10H2O → Na2CO3. 10H2O
Uses
(i) Used in glass, soap, and paper industries.
(ii) Employed in the manufacture of sodium compounds such as borax.
(iii) As a household cleansing agent.
(iv) In paper and soap/detergent industries.
(v) For the softening of water.
(vi) A mixture of NaCO3 and K2CO3 is used as a fusion mixture.
(vii) In the textile industry and petroleum refining.
Acids Bases and Salts Class 10 Notes
Baking Soda
(i) Chemical formula: NaHCO3
(ii) It is produced on a large scale by treating a cold and concentrated solution of sodium chloride (brine) with ammonia and carbon dioxide.
NaCl + H2O + CO2 + NH3 → NH4Cl + NaHCO3
(iii) On heating, it decomposes to give sodium carbonate with the evolution of carbon dioxide.
2NaHCO3 → Na2CO3 + H2O + CO2
Uses
(i) Used as an antacid to treat acidity of the stomach.
(ii) Used to make baking powder, which is used in the preparation of cakes, bread, etc.
(iii) Used in soda-acid fire extinguishers.
Bleaching Powder
(i) Bleaching powder is manufactured from chlorine gas.
(ii) It is produced by the action of chlorine on dry slaked lime [Ca(OH)2].
Ca(OH)2 + Cl2 → CaOCl2 + H2O
(iii) It is represented as CaOCl2
Uses
(i) For bleaching cotton and linen in the textile industry and for bleaching wood pulp in the paper industry.
(ii) Used for disinfecting drinking water to make it free of germs.
Plaster of Paris
It is prepared by heating gypsum at 373 K. On heating, it loses water molecules and becomes calcium sulphate hemihydrate (CaSO4.1/2 H2O) which is called Plaster of Paris.
CaSO4.2H2O → CaSO4.½ H2O + 1½ H2O
Gypsum Plaster of Paris
Uses
(i) Used in hospitals as plaster for supporting fractured bones in the right position.
(ii) Used as a fire-proofing material.

The water of Crystallisation
(i). Water molecules that form a part of the structure of a crystal are called water of crystallization.
(ii). The salts which contain water of crystallization are called hydrated salts.
(iii) Every hydrated salt has a fixed number of molecules of crystallization in its one formula unit. For Example CuSO4.5H2O, Na2CO3.10H2O, CaSO4.5H2O, and FeSO4.7H2O.
Acids Bases and Salts Class 10 Notes