Biomolecules Class 12 Important Questions
Que1. How many asymmetric carbon atoms are present in D (+) glucose?
Ans 1. Four asymmetric carbon atoms are present in D (+) glucose.

Que 2. What are the two end products of cellular respiration?
Ans 2. These are CO2 and H2O.
Que 3. Give the significance of (+)-sign in the name D- (+)-glucose.
Ans 3. (+) sign indicates the dextrorotatory nature of glucose.
Que 4. Give the significance of the prefix ‘D’ in the name D- (+)-glucose.
Ans 4. ‘D’ Signifies that –OH group on C-5 is on the right-hand side.
Que 5. Describe the term D-and L-configuration used for amino acids with examples
Ans 5. Consider the following configurations (I and II) of a-amino acids.
NH2 group on the α-carbon oriented toward left (as in structure I) is referred to as L-amino acid and NH2 group oriented toward the right (as in structure II) is referred to as D-amino acid.

Biomolecules Class 12 Important Questions
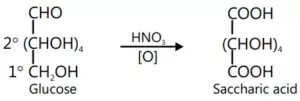
Que 6. How will you distinguish 1° and 2° hydroxyl groups present in glucose? Explain with reactions.
Ans 6. The –OH group on the terminal carbon atom (i.e. C6 ) is called the 1° hydroxyl while the rest of the four remaining OH groups present on C2, C3, C4, and C5 are called 2° hydroxyl groups. While 1° Hydroxyl groups are easily oxidized to give carboxylic acids but 2° hydroxyl groups undergo oxidation only under drastic conditions. For example, glucose on oxidation with HNO3 gives a dicarboxylic acid, saccharic acid having the same number of carbon atoms as glucose. This indicates that glucose contains one 1° hydroxyl group while the remaining four are 2° hydroxyl groups.
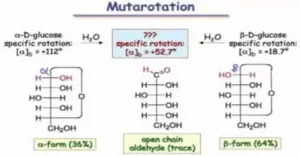
Que 7. What is mutarotation?
Ans 7. Mutarotation is the change in the optical rotation because of the change in the equilibrium between two anomers.
Que 8. Name the molecule which provides energy for all the activities of a cell.
Ans 8. Adenosine triphosphate (ATP).
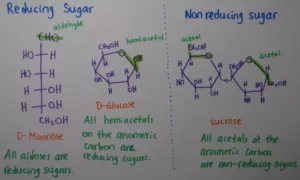
Que 9. What is the difference between reducing and non‐reducing sugars or carbohydrates?
Ans 9. All those carbohydrates which contain aldehydic and ketonic group in the hemiacetal or hemiketal form and reduce Tollen’s reagent or Fehling’s solution are called reducing carbohydrates while others that do not reduce these reagents are called non‐reducing sugars.
Biomolecules Class 12 Important Questions
Que 10. What is the enzyme present in saliva? What is its function?
Ans 10. The enzyme present in the saliva is amylase. It converts starch into maltose.
Que 11. What are enzymes?
Ans 11. Most of the reactions in living systems proceed at very high rates under physiological pH of 7.4 and a temperature of 37 0C under one atm pressure. All these reactions are catalyzed by a group of biomolecules called enzymes. Thus enzymes are biological catalysts, chemically almost all the enzymes are globular proteins.
Que 12. What are carbohydrates? Why they are generally optically active?
Ans 12. Hydrocarbons are the hydrates of carbon having the general formula Cx(H2O)y. Carbohydrate molecules have one or more asymmetric carbon atoms. Hence they are generally optically active.
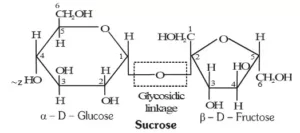
Que 13. Define glycosidic linkage?
Ans 13. The two monosaccharide units are joined together through an ethereal or oxide linkage formed by the loss of a molecule of H2O. Such a linkage between two monosaccharide units through oxygen atoms is called glycosidic linkage.
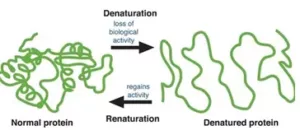
Que 14. What do you understand by denaturation of proteins?
Ans 14. When a protein in its native form, is subjected to physical change like in temperature or chemical change like change in pH, the hydrogen bonds are disturbed. Due to this, globules unfold and helixes get uncoiled and protein loses its biological activity. This is called denaturation of the protein.
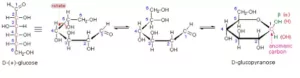
Que 15. Despite having an aldehyde group glucose does not give 2, 4-DNP test. What does this indicate? What is the significance of D and (+) in a-D-(+) Glucopyranose.
Ans 15. Glucose does not have an open structure and hence it does not have a free –CHO group. Actually –CHO group combines with C5 –OH to form a hemiacetal. Thus glucose largely exists in cyclic hemiacetal form along with a very small amount (< 0.5 %) of the open-chain structure. Since the concentration of open-chain form is low and its reaction with 2, 4-DNP is reversible, therefore the formation of 2,4-DNP derivative can not disturb the equilibrium to generate more of the open-chain form from the cyclic hemiacetal form, and hence it does not react with 2, 4-DNP. The capital letter D in D-(+)-Glucopyranose indicates that the C-5 –OH group is oriented towards the right, while the sign (+) shows that glucopyranose is dextrorotatory.
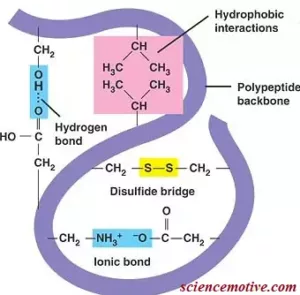
Que 16. What type of bonding occurs in globular protein?
Ans 16. Globular protein is spherical in nature and is water-soluble. Globular protein may have the following types of bonding: hydrogen bonding, disulphide bridges, ionic or salt bridges, and hydrophobic interactions.
Biomolecules Class 12 Important Questions
Que 17. Explain the functions of nucleic acids.
Ans 17. Nucleic acids are large biomolecules consisting of RNA and DNA. Nucleic acids have two important functions:
i) Replication: Due to its unique property of self-replication (the process by which a single DNA molecule produces two identical species of itself), it is responsible for maintaining the heredity traits from one generation to another.
ii) Protein synthesis: RNA helps in the biosynthesis of proteins and is, in one way, responsible for the process of learning and memory storage. Furthermore, it sends information and instructions to the cell for the manufacture of specific proteins.
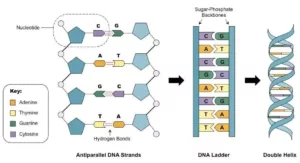
Que 18. The two strands of DNA are not identical, but are complimentary’. Explain this statement.
Ans 18. DNA consists of two strands of polynucleotides coiled around each other in the form of a double helix. The nucleotides making up each strand of DNA are connected by phosphate ester bonds. This forms the backbone of each DNA strand, from which the bases extend. The bases of one strand of DNA are paired with the bases on the other strand by means of hydrogen bonding. This hydrogen bonding is very specific as the structure of bases permits only one mode of pairing. Adenine pairs only thymine via two hydrogen bonds and guanine pairs with cytosine through three hydrogen bonds. The two strands of DNA are said to be complementary to each other in the sense that the sequence of bases in one strand automatically determines that of the other. These strands are not identical.
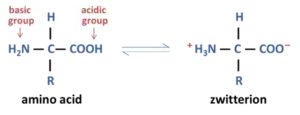
Que 19. What is zwitterion?
Ans 19. Amino acids are usually colorless, crystalline solids. These are water-soluble, high melting solids and behave like salts rather than simple amines or carboxylic acids. This behavior is due to the presence of both acidic (carboxylic group) and basic (amino group) groups in the same molecule. In an aqueous solution, the carboxyl group can lose a proton, an amino group can accept a proton, giving rise to a dipolar ion known as a zwitterion.
Que 20. What is the effect of pH on the action of an enzyme?
Ans 20. The low or high pH values can cause denaturation of the protein and hence make the enzyme’s protein inactive.
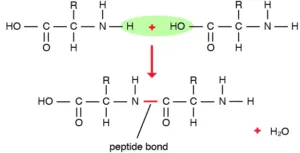
Que 21. What is a peptide bond?
Ans 21. The covalent bond NH – CO formed between –NH2 group of one amino acid and –COOH group of the other with the elimination of a molecule of water is called peptide bond or peptide linkage. Amino acids are joined together, by peptide bonds.
Biomolecules Class 12 Important Questions
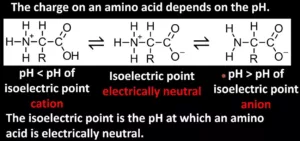
Que 22. What is the isoelectric point?
Ans 22. The pH at which there is no net migration of the amino acid under the influence of an applied electric field is called the isoelectric point. For example, isoelectric point of glycine is 6.1.
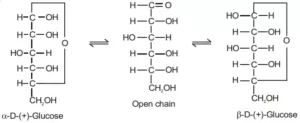
Que 23. An optically active compound having molecular formula C6H12O6 exists in two cyclic forms A and B. When A and B are dissolved in water they show the following equilibrium.
![]()
What are such isomers called?
Ans 23. The optically active compound having molecular formulae C6H12O6 is called glucose. It exists in two stereo-isomeric forms A and B. These two isomers which differ in a configuration only at the anomeric (first) carbon are called anomers.
a-forms is less stable than b-form therefore equilibrium mixture contains 36% of a-form [(a) D = 111º] and 64% of B-form [(a) D = 19.20].
Que 24. Why are carbohydrates generally optically active?
Ans 24. It is due to the presence of Chiral Carbon atoms in their molecules.
Que 25. Differentiate between DNA and RNA.
Ans 25.

Que 26. (i) What products would be formed when a nucleotide from DNA containing thymine is hydrolyzed?
(ii)How will you distinguish 1° and 2° hydroxyl groups present in glucose?
Ans 26. (i) Complete hydrolysis of DNA yields pentose sugar, phosphoric acid, and thymine.
(ii) On oxidation with nitric acid, glucose well as gluconic acid both yield a dicarboxylic acid, saccharic acid. This indicates the presence of a primary alcoholic (–OH) group in glucose.
Que 27. Explain the tertiary structure of Protein.
Ans 27. The tertiary structure of proteins: The tertiary structure of proteins represents overall folding of the polypeptide chains i.e., further folding of the secondary structure. It gives rise to two major molecular shapes viz. fibrous and globular. The main forces which stabilize the 2° and 3° structures of proteins are hydrogen bonds, disulphide linkages, van der Waals and electrostatic forces of attraction.
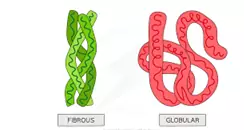
Que 28. How is a globular protein different from fibrous protein?
Ans 28.

Biomolecules Class 12 Important Questions
Que 29. Name one fibrous and one globular protein each.
Ans 29. Fibrous – Keratin
Globular – Haemoglobin
Que 30. What is invert sugar?
Ans 30. An equimolar mixture of glucose and fructose obtained by hydrolysis of sucrose in presence of an acid such as dil. HCl or enzyme invertase is called invert sugar.
Que 31. Differentiate between essential and non-essential amino acids.
Ans 31. Essential amino acids are needed for vital life processes and can only be obtained through foods. They are nine in number and are present in proteinous foods either plant e.g beans and nuts or animal e.g dairy, cottage cheese, fish, meat, turkey, poultry, etc. They are methionine, isoleucine, lysine, leucine, histidine, phenylalanine, threonine, tryptophan, and valine.
On the other hand, non-essential amino acids can be produced by the body and are eleven in number. However, during sickness, the body may not be able to produce enough and amino acid supplements may be required according to the doctor’s prescription. Non-essential amino acids include arginine, glycine, alanine, serine, aspartic acid, glutamic acid, cysteine, cysteine, proline, alanine, and tyrosine.
Biomolecules Class 12 Important Questions



