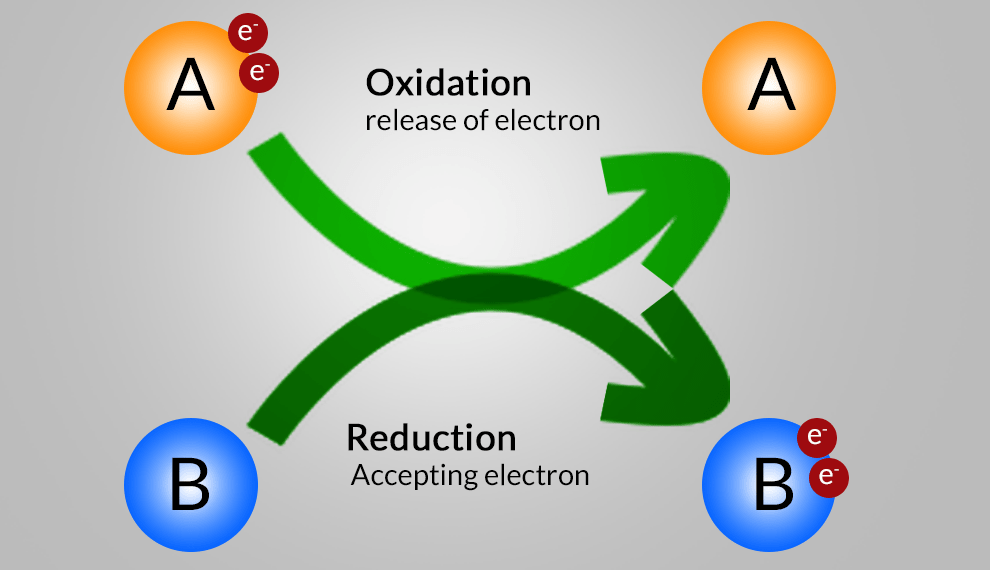
Redox Reaction, Oxidation and Reduction Reactions with exact equations This part of the types of chemical reaction of chapter Chemical Reactions and Equations involve the types Redox Reaction, Oxidation and Reduction Reactions with exact equations. v). Redox Reaction: A reaction in which reduction and oxidation takes place simultaneously. ZnO (s) + C (s) → Zn (s) + CO (g) Zinc Carbon Zinc Carbon Oxide Monoxide vi). Oxidation Reaction: Oxidation is the gain of oxygen. For example: 2…
Author: Dr. Vikas Jasrotia
Combination, Decomposition, Displacement And Double Displacement reactions Types of Chemical Reactions: i). Combination Reaction ii). Decomposition Reaction a). Thermal Decomposition b). Electrolytic Decomposition iii). Displacement Reaction OR Substitution reaction iv). Double Displacement Reaction a). Precipitation Reaction b). Neutralisation Reaction v). Redox Reaction vi). Oxidation Reaction vii). Reduction Reaction viii). Endothermic Reaction ix). Exothermic Reaction Explanation of the types of Chemical reactions i.e Combination, Decomposition, Displacement And Double Displacement reactions using examples: i). Combination Reaction: The reactions in which two or more substances combine to…
Chemistry Balancing Chemical Equations Practice Balanced Equation A balanced equation is one in which the number of atoms on the reactant and product sides is equal. Or A number of atoms of an element on the reactant side = number of atoms of that element on the product side. The simple form of representation of a chemical reaction in words is known as a word equation. Magnesium + Oxygen ———–> Magnesium Oxide Representation of a chemical reaction with the help of chemical formula is called a chemical equation. 2Mg + O2→ 2MgO Chemistry Balancing Chemical…
Chemical Reactions And Equations Notes Physical Change Change in physical properties. Melting Boiling Condensation Note- No change occurs in the identity of the substance Chemical Change Atoms in the reactants are rearranged to form one or more different substances. Old bonds are broken, new bonds are formed. Reactants lose their properties to form products of different properties. Examples – Cooking of food Rusting of iron Heating of Lead nitrate Souring of milk Ripening of fruit. Rusting of iron is a chemical change because A new substance iron oxide is formed. The change is permanent; the article has got a rust layer (which may only peal off). There is an increase in mass when rust forms. An energy change…