Aldehydes and Ketones – Distinguish Tests Class 12
Aldehydes and Ketones
Que. Give simple chemical tests to distinguish between the following pairs of compounds.
(i) Propanal and propanone
(ii) Acetophenone and benzophenone
(iii) Phenol and benzoic acid
(iv) Benzoic acid and ethyl benzene
(v) Ethanal and propanal
(vi) Benzaldehyde and acetophenone
Answers:
(i) Propanal and Propanone: These two are distinguished by the iodoform test. propanal does not give an iodoform test when it reacts with I2 in the presence of NaOH while propanone gives an iodoform test when reacts with I2 in the presence of NaOH.
CH3COCH3 + NaOI → CH3COONa + CHI3 + NaOH
(ii) Acetophenone and Benzophenone: Acetophenone gives a positive iodoform test whereas benzophenone (C6H5COC6H5) does not.
C6H5COCH3 + NaOI → C6H5COONa + CHI3 + NaOH
Yellow ppts
C6H5COC6H5 + NaOI → No Yellow ppts
(iii) Phenol and Benzoic acid: Phenol gives a violet coloration with neutral FeCl3 solution while benzoic acid gives a buff coloured precipitate of ferric benzoate.
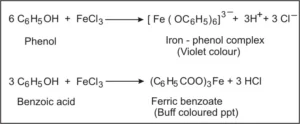
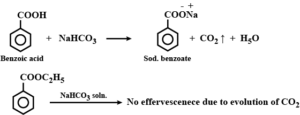
(iv) Benzoic acid and Ethylbenzene: Benzoic acid on warming with sodium hydrogen carbonate gives brisk effervescence of CO2 gas while ethyl benzoate does not respond to this test.
(v) Ethanal and propanal: Aldehydes and ketones having at least one methyl group linked to the carbonyl carbon atom respond to the iodoform test. Ethanal having one methyl group linked to the carbonyl carbon atom responds to this test. But propanal does not have a methyl group linked to the carbonyl carbon atom and thus, it does not respond to this state.
CH3CHO + 4 NaOH + 3 I2 → CHI3 (Yellow ppt.) + HCOONa + 3 NaI + 3 H2O
Propanal → Does not give this test
(vi) Benzaldehyde and Acetophenone: Benzaldehyde is an aromatic aldehyde, while acetophenone is an aromatic ketone compound. Therefore, these can be distinguished by using Tollen’s reagent because the aldehyde functional group can form a precipitate with this reagent. Benzaldehyde can reduce Tollen’s reagent, giving a red-brown precipitate of Cu2O, whereas acetophenone does not show any reaction towards Tollen’s reagent.
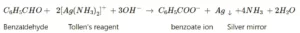
Aldehydes and Ketones



