Ncert Solutions for Class 12 Chemistry Chapter 1
Exercise Solutions
The Solid State
Que 1. Define the term ‘amorphous’. Give a few examples of amorphous solids.
Ans 1: A solid is said to be amorphous if the constituent particles are not arranged in any regular fashion. They may have only short-range order. Amorphous solids are generally obtained when the melts are rapidly cooled, e.g., glass, plastics, amorphous silica, etc.
Que 2. What makes a glass different from a solid such as quartz? Under what conditions could quartz be converted into glass
Ans 2: Glass is an amorphous solid in which the constituent particles (SiO4 tetrahedral) have only a short-range order and there is no long-range order. In quartz, the constituent particles (SiO4 tetrahedral) have both short-range as well as long-range orders. On melting quartz and then cooling it rapidly, it is converted into glass.
Que 3: Classify each of the following solids as ionic, metallic, molecular, network (covalent) or amorphous. Tetraphosphorus decoxide, P4O10, Ammonium phosphate, (NH4)3PO4, SiC, l2, P4, Plastic, Graphite, Brass, Rb, LiBr, Si.
Ans 3: P4O10 – molecular, (NH4)3PO4 – ionic, SiC – network (covalent), I2 – molecular, P4 – molecular, plastic – amorphous, graphite – covalent, brass – metallic, Rb – metallic, LiBr – ionic, Si – covalent
Que 4. (i) What is meant by the term ‘coordination number’?
(ii) What is the coordination number of atoms:
(a) in a cubic close-packed structure?
(b) in a body-centred cubic structure?
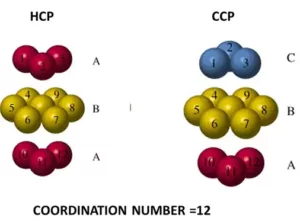
Ans 4: (i) Coordination number is defined as the number of nearest neighbours in a close packing. In ionic crystals, the coordination number of an ion in the crystal is the number of oppositely charged ions surrounding that particular ion.
(ii) (a) Coordination number of atoms in a cubic close-packed structure is 12.
(b) Coordination number of atoms in a body-centred cubic structure is 8.
Que 5. How can you determine the atomic mass of an unknown metal if you know its density and the dimension of its unit cell? Explain.
Ans 5.

Ncert Solutions for Class 12 Chemistry Chapter 1
Que 6. The stability of a crystal is reflected in the magnitude of its melting point. Comment. Collect melting points of solid water, ethyl alcohol, diethyl ether and methane from a data book. What can you say about the intermolecular forces between these molecules?
Ans 6. The melting points of some compounds are given below
Water = 273 K, Ethyl alcohol = 155.7 K, Diethyl ether = 156.8 K, Methane = 90.5 K.
The higher the melting point, the stronger are the forces holding the constituent particles together and hence greater is the stability.
The intermolecular forces in water and ethyl alcohol are mainly hydrogen bonding. The higher melting point of water as compared to alcohol shows that hydrogen bonding in ethyl alcohol molecules is not as strong as in water molecules. Diethyl ether is a polar molecule. The intermolecular forces present in them are the dipole-dipole attraction. Methane is a non-polar molecule. The only forces present in them are the weak van der Waals forces (London or Dispersion Forces).
Que 7. How will you distinguish between the following pairs of terms?
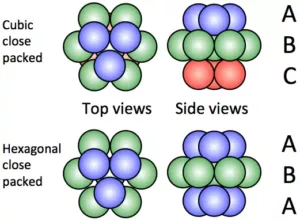
(a) Hexagonal close-packing and cubic close-packing
(b) Crystal lattice and unit cells
(c) Tetrahedral void and octahedral void
Ans 7. (a) In Hexagonal Close Packing (hcp), the spheres of the third layer are vertically above the spheres of the first layer (ABABAB….type). On the other hand, in cubic close packing (ccp), the spheres of the fourth layer are present above the spheres of the first layer (ABCABC…..type).
(b) Crystal lattice: It depicts the actual shape as well as the size of the constituent particles in the crystal. It is, therefore, called space lattice or crystal lattice.
Unit cell: Each brick represents the unit cell while the block is similar to space or crystal lattice. Thus, a unit cell is the fundamental building block of the space lattice.

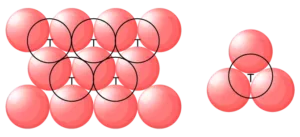
(c) Tetrahedral void: A tetrahedral void is formed when a triangular void made by three spheres of a particular layer and touching each other.
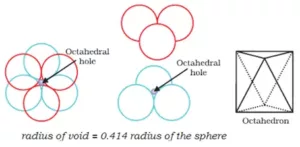
Octahedral void: An octahedral void or site is formed when three spheres arranged at the corners of an equilateral triangle are placed over another set of spheres.
Que 8. How many lattice points are there is one unit cell of each of the following lattices?
(i) Face centred cubic
(if) Face centred tetragonal
(iii) Body Centred Cubic
Ans 8. (i) There are 14 (8 from the corners + 6 from the faces) lattice points in face-centred cubic.
(ii) There are 14 (8 from the corners + 6 from the faces) lattice points in face-centred tetragonal.
(iii) There are 9 (1 from the centre + 8 from the corners) lattice points in body-centred cubic.
Que 9. Explain
(i) The basis of similarities and differences between metallic and ionic crystals.
(ii) Ionic solids are hard and brittle.
Ans 9. (i) Metallic and ionic crystals
(a) There is an electrostatic force of attraction in both metallic and ionic crystals.
(b) Both have high melting points.
(c) Bonds are non-directional in both cases.
Differences:
(a) Ionic crystals are bad conductors of electricity in the solids state as ions are not free to move. They can conduct electricity only in a die molten state or in an aqueous solution. Metallic crystals are good conductors of electricity in the solid-state as electrons are free to move.
(b) Ionic bond is strong due to strong electrostatic forces of attraction.
The metallic bond may be strong or weak depending upon the number of valence electrons and the size of the kernels.
(ii) Ionic solids are hard and brittle. Ionic solids are hard due to the presence of strong electrostatic forces of attraction. The brittleness in ionic crystals is due to the non-directional bonds in them.
Que 10. Calculate the efficiency of packing in the case of a metal crystal for
(i) Simple Cubic
Answer: Let the side of a simple cubic lattice is ‘a’ and the radius of the atom present in it is ‘r’.
Since, edges of atoms touch each other, therefore, a = 2r (for simple cubic lattice)
Volume of cube =Side3 = a3 = (2r)3
The volume of one atom =4/3πr3

(ii) Body-Centred Cubic:

Ncert Solutions for Class 12 Chemistry Chapter 1
Que 11. Silver crystallises in fcc lattice. If the edge length of the cell is 4.07 x 10-8 cm and density is 10.5 g cm-3, calculate the atomic mass of silver.
Ans 11. It is given that the edge length, a = 4.077 × 10−8 cm
Density, d = 10.5 g cm−3
As the lattice is fcc type, the number of atoms per unit cell, z = 4
We also know that NA = 6.022 × 1023 mol−1
Using the relation:
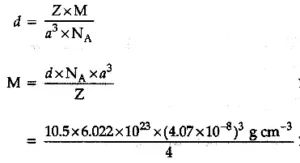
= 107.09 g/mol
Que 12. A cubic solid is made of two elements P and Q. Atoms of Q are at the corners of the cube and P at the body-centre. What is the formula of the compound? What is the coordination number of P and Q?
Ans 12. It is given that the atoms of Q are present at the corners of the cube.
Therefore, the number of atoms of Q in one unit cell = 8 x (1/8) = 1
It is also given that the atoms of P are present at the body-centre.
Therefore, the number of atoms of P in one unit cell = 1
This means that the ratio of the number of P atoms to the number of Q atoms, P:Q = 1:1
Hence, the formula of the compound is PQ.
The coordination number of both P and Q is 8.
Que 13. Niobium crystallises in a body-centred cubic structure. If the density is 8.55 g cm–3, calculate the atomic radius of niobium using its atomic mass of 93 u.
Ans 13. Given, density (d) = 8.55 g cm-3
Atomic Mass (M) = 93 u = 93 g mol-1
Atomic radius (r) = ?
We know, Avogadro Number NA = 6.022 × 1023mol-1
Since the given lattice is bcc
Hence, the number of atoms per unit cell (z) = 2

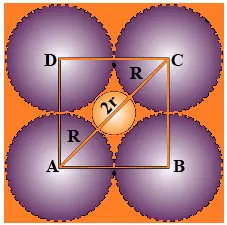
Que 14. If the radius of the octahedral void is r and the radius of the atoms in close-packing is R, derive the relation between rand R.
Ans 14. A sphere is fitted into the octahedral void as shown in the diagram.

Que 15. Copper crystallises into a fee lattice with an edge length of 3.61 x 10-8 cm. Show that the calculated density is in agreement with its measured value of 8.92 g/cm3.
Ans 15. Given, edge length (a) = 3.61 × 10-8cm
Hence, the number of atoms per unit cell in fcc (z) = 4
Avogadro’s Number (NA) = 6.022 ××1023 g mol-1
To prove, density (d) = 8.92 g cm-3
We know that density (d) = zM/a3NA

Ncert Solutions for Class 12 Chemistry Chapter 1
Que 16. The analysis shows that nickel oxide has the formula Ni0.98 O1.00. What fractions of nickel exist as Ni2+ and Ni3+ ions?
Ans 16. Given, the formula of nickel oxide = Ni0.98O1.00
So, Ni : O = 0.98 : 1.00 = 98 : 100
Total charge on O2- = 100 × (-2) = -200
Now, let number of Ni2+ = x
So, number of Ni3+ = 98 – x
Since the compound is neutral
Hence, number of Ni2+ ions + number of Ni3+ ions + number of O2- ions = 0
= x(−2) + (98−x) × (−3) + (−200) = 0
= −2x – 294 + 3x – 200 = 0
= x – 94 = 0
x = 94
So, number of Ni2+ ions = 94
Number of Ni3+ ions = 98 – 94 = 4
Now, fraction of nickel existing as Ni2+ ions = 94/98 = 0.959
Fraction of nickel existing as Ni3+ = 4/98 = 0.041
Que 17. What are semi-conductors? Describe the two main types of semiconductors and contrast their conduction mechanisms.
Ans 17. Semiconductor: – Solids having an intermediate range of conductivity, i.e. from 10–6 to 104 ohm–1 m–1 are called semiconductors. Semiconductors are of the following two types:
(a) n-type of semiconductors
(b) p-type of semiconductors
(a) n-type semiconductors – An n-type semiconductor is an intrinsic semiconductor doped with phosphorus (P), arsenic (As), or antimony (Sb) as an impurity. Silicon of Group IV has four valence electrons and phosphorus of Group V has five valence electrons. If a small amount of phosphorus is added to a pure silicon crystal, one of the valence electrons of phosphorus becomes free to move around (free-electron) as a surplus electron. When this free electron is attracted to the “+” electrode and moves, the current flows.
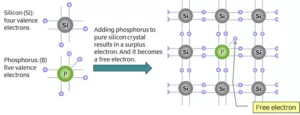
(b) p – type of semiconductors – The extrinsic p–Type Semiconductor is formed when a trivalent impurity is added to a pure semiconductor in a small amount, and as a result, a large number of holes are created in it. A large number of holes are provided in the semiconductor material by the addition of trivalent impurities like Gallium and Indium.
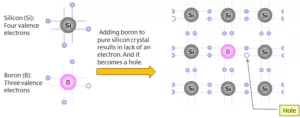
Que 18. Non-stoichiometric cuprous oxide, Cu2O can be prepared in the laboratory. In this oxide, the copper to oxygen ratio is slightly less than 2:1. Can you account for the fact that this substance is a p-type semiconductor?
Ans 18. When cuprous oxide is prepared in the laboratory; the ratio of copper to the oxygen in the compound becomes slightly less than 2:1. This happens because some of the Cu+ ions are replaced by the Cu2+ ions. In this process, one Cu2+ ion replaces two Cu+ ions. As two Cu+ ions are replaced by one Cu2+ ion, this creates defects because of creating vacant space, i.e. positive holes.
Because of the creation of holes due to this defect; this compound conducts electricity through these positive holes.
As semiconductors that are formed by electron-deficient impurities are called p-type semiconductors; thus, cuprous oxides so formed in the laboratory are the p-type of semiconductors.
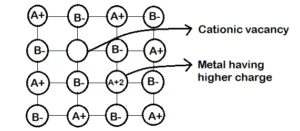
Que 19. Ferric oxide crystallises in a hexagonal dose- packed array of oxide ions with two out of every three octahedral holes occupied by ferric ions. Derive the formula of the ferric oxide.
Ans 19. Given, Ferric oxide crystallizes in a hexagonal close-packed array of oxide ions.
Two out of every three octahedral holes are occupied by ferric ions.
Let the number of oxide ions = x
Therefore the number of octahedral voids = x
Since, two out of every three octahedral holes are filled by ferric ions,
Thus, voids filled by ferric ions
Fe3+ = 2/3x
Therefore, the number of ferric ions
Fe3+ = 2/3x
Now, the ratio of ferric ions to the oxide ions
i.e. Fe3+ : O2- =2/3x : x
= 2 : 3 Hence, the formula of ferric oxide is Fe2O3
Que 20. Classify each of the following as being either a p-type or n-type semiconductor:
i). Ge doped with In
ii). B doped with Si.
Ans 20. i). Ge is a group 14 element and In is a group 13 element. Hence, an electron-deficient hole is created and therefore, it is a p-type semiconductor.
ii). Boron is a group 13 element and Si is a group 14 element, there will be a free electron, So, it is an n-type semiconductor.
Ncert Solutions for Class 12 Chemistry Chapter 1
Que 21. Gold (atomic radius = 0.144 nm) crystallises in a face-centred unit cell. What is the length of the side of the unit cell?
Ans 21. Given, atomic radius = 0.144 nm
Type of unit cell = face centred
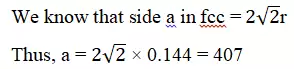
Thus side of the given cell = 407 nm
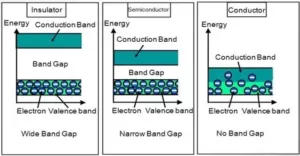
Que 22. In terms of band theory, what is the difference
(i) Between a conductor and an insulator
(ii) Between a conductor and a semiconductor?
Ans 22. In most of the solids and in many insulating solids conduction takes place due to the migration of electrons under the influence of the electric field. However, in ionic solids, it is the ions that are responsible for conducting behaviour due to their movement.
(i) Between a conductor and an insulator: In conductors there is no energy gap between the valence band, which facilitates the flow of electrons easily under an applied electric field and metals show conductivity. While in insulators there is a large energy gap between the valence band and electrons cannot jump to it i.e. large energy gap prevents the flow of electricity.
(ii) Between a conductor and a semiconductor: In conductors there is no energy gap between the valence band and conduction band, which facilitates the flow of electrons easily under an applied electric field and metals show conductivity.
While in semiconductors, there is a small energy gap between the valence bond and the conduction band. The small gap between bands facilitates some electrons to jump to the conduction band by acquiring extra energy.
Que 23. Explain the following terms with suitable examples :
(i) Schottky defect
(ii) Frenkel defect
(iii) Interstitial defect
(iv) F-centres
Ans 23. (i) Schottky defect: Schottky defect is basically a vacancy defect. In this defect, an equal number of cations and anions are missing to maintain electrical neutrality. It decreases the density of a substance. It is a common defect in ionic compounds of high coordination number where both cations and anions are of the same size, e.g., KCl, NaCl, KBr, etc.
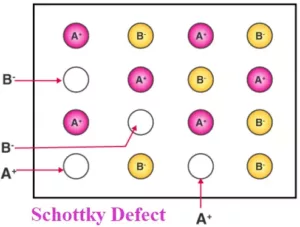
(ii) Frenkel defect: This defect arises when some of the ions (generally cation due to its small size) in the lattice occupy interstitial sites leaving lattice sites vacant. This defect is generally found in ionic crystals where anion is much larger in size than the cation, e.g., AgBr, ZnS, etc. Due to this defect density does not change.
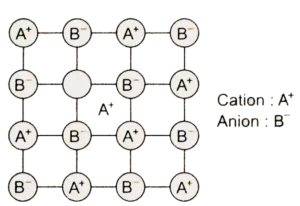
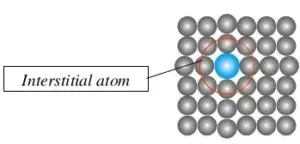
(iii) Interstitial defect: Sometime in the formation of lattice structure some of the atoms or ions occupy the vacant interstitial site, and are known as interstitials. These interstitials are generally small size non-metals, such as H, B, C, etc. The defect that arises because of interstitials is called an interstitial defect.
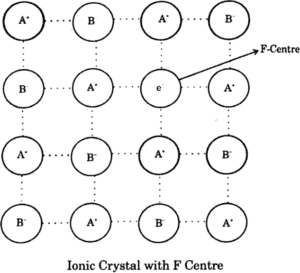
(iv) F-centres: This is a type of defect and called a metal excess defect. These type of defects seen because of the missing of anions from the regular site leaving a hole which is occupied by an electron to maintain the neutrality of the compound. The hole occupied by the electron is called F-centre and responsible for showing colour by the compound.
Que 24. Aluminium crystallises in a cubic close-packed structure. Its metallic radius is 125 pm.
(i) What is the length of the side of the unit cell?
(ii) How many unit cells are there in 1.00 cm3 of aluminium?
Ans 24. Given, the radius of an atom (r) = 125 pm
(i) For ccp structure, we know that
Where, r = radius and a = length of side

Que 25. If NaCI is doped with 10-3 mol % SrCl2, what is the concentration of cation vacancies?
Ans 25. Let moles of NaCI = 100
∴ Moles of SrCl2 doped = 10-3
Each Sr2+ will replace two Na+ ions. To maintain electrical neutrality it occupies one position and thus creates one cation vacancy.
∴ Moles of cation vacancy in 100 moles NaCI = 10-3
Moles of cation vacancy in one mole
NaCI = 10-3 × 10-2 = 10-5
∴ Number of cation vacancies
= 10-5 × 6.022 × 1023 = 6.022 × 1018 mol-1.
Que 26. Explain the following with suitable example:
(i) Ferromagnetism
(ii) Paramagnetism
(iii) Ferrimagnetism
(iv) Antiferromagnetism
(v) 12-16 and 13-15 group compounds.
Ans 26. (i) Ferromagnetism: Substances that are attracted strongly with magnetic field are called ferromagnetic substances, such as cobalt, nickel, iron, gadolinium, chromium oxide, etc. Ferromagnetic substances can be permanently magnetized also. In a solid-state, the metal ions of ferromagnetic substances are grouped together into small regions called domains and each domain acts as a tiny magnet.
(ii) Paramagnetism: Substances that are attracted slightly by the magnetic field and do not retain the magnetic property after removal of the magnetic field is called paramagnetic substances. They are magnetized in the same direction as that of the applied field. This property is shown by those substances whose atoms, ions or molecules contain unpaired electrons For example O2-, Cu2+, Fe3+, Cr3+, Magnesium, molybdenum. Substances that are slightly attracted in the magnetic field and in which domains are grouped in parallel and anti-parallel direction but in unequal number are called ferromagnetic substances and this property is called ferrimagnetism. Examples include Fe3O4 (magnetite), ferrites such as MgFe2O4 and ZnFe2O4.
(iv) Antiferromagnetism: Substances in which domain structure is similar to ferromagnetic substances but are oriented oppositely, which cancel the magnetic property are called antiferromagnetic substances and this property is called antiferromagnetism. For example; MnO.

(v) 12-16 and 13-15 group compounds: When the solid-state materials are produced by the combination of elements of groups 13 and 15, the compounds thus obtained are called 13-15 compounds. For example, InSb, AlP, GaAs, etc.
Combination of elements of groups 12 and 16 yields some solid compounds which are referred to as 12-16 compounds. For example, ZnS, CdS, CdSe, HgTe, etc. In these compounds, the bonds have an ionic character.
Ncert Solutions for Class 12 Chemistry Chapter 1



