MCQs on The p-Block Elements Class 12 Chemistry
MCQs on The p-Block Elements Class 12 Chemistry
Que 1. Each of the following is true for white and red phosphorus except that they
(a) Are both soluble in CS2
(b) Can be oxidised by heating in air
(c) Consists of the same kind of atoms
(d) Can be converted into one another
Ans 1: (a) White phosphorus is soluble in CS2 whereas red phosphorus is insoluble in it.
Que 2. Which of the following is a tetrabasic acid
(a) Orthophosphorus acid (b) Orthophosphoric acid
(c) Metaphosphoric acid (d) Pyrophosphoric acid
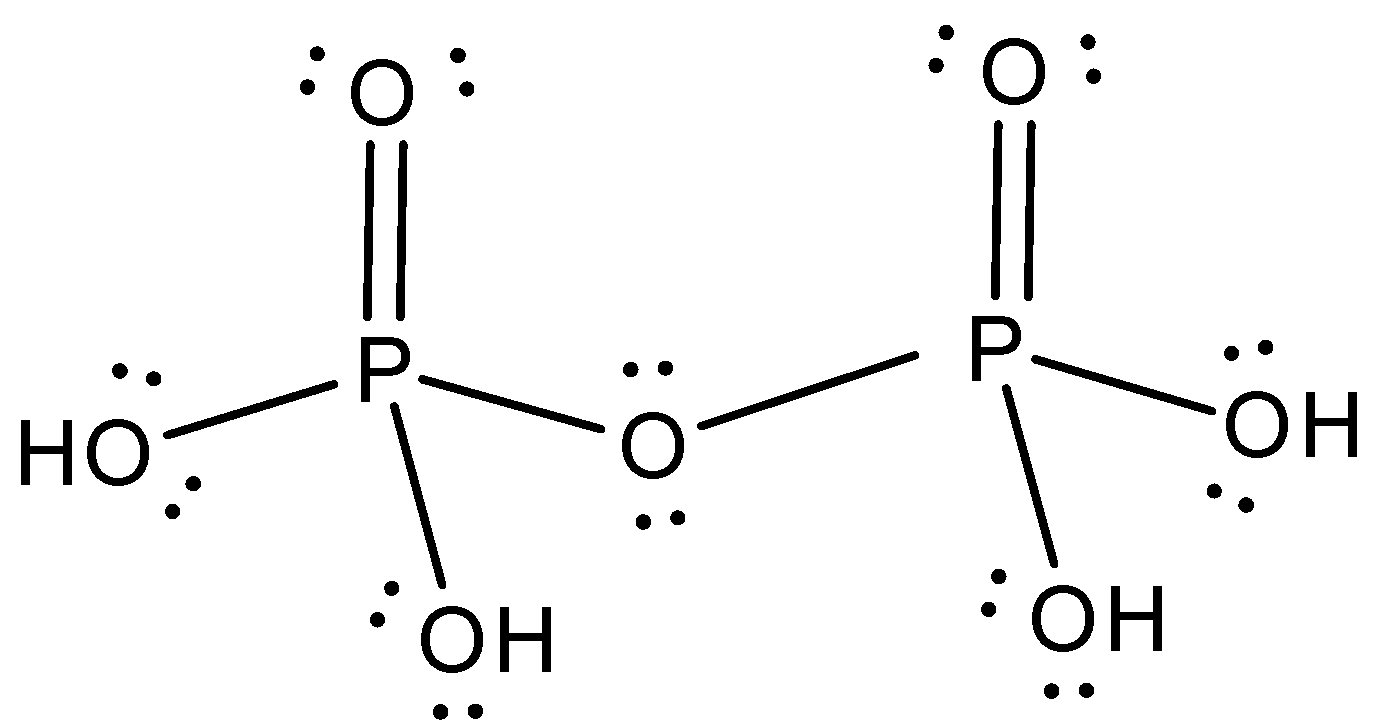
Ans 2: (d) H4P2O7 pyrophosphoric acid
Tetrabasic ( 4 – OH groups)
Que 3. Phosphine is prepared by the reaction of
(a) P and H2SO4 (b) P and NaOH
(c) P and H2S (d) P and HNO3
Ans 3: (b) P4 + NaOH + H2O → PH3 + 3NaH2PO2
Que 4. When concentrated nitric acid is heated, it decomposes to give
(a) O2 and N2 (b) NO
(c) N2O5 (d) NO2 and O2
Ans 4: (d) 4HNO3 → 4NO2 + O2 + 2H2O
Que 5. The element which catches fire in the air at 300C and is stored underwater is
(a) Calcium (b) Sodium
(c) Phosphorus (d) Zinc
Ans 5: c) Because of its very low ignition temperature (303K) it is always kept underwater.
MCQs on The p-Block Elements Class 12 Chemistry
Que 6. A solution of ammonia in water contains
(a) H+ (b) OH–
(c) Only NH4+ (d) OH–, NH4+ and NH4OH molecules
Ans 6: (d) NH3 when dissolved in water forms
NH3 + H2O → NH4+ + OH– ⇌ NH4OH
Que 7. Which of the following halides is most acidic
(a) PCl3 (b) BiCl3
(c) SbCl3 (d) CCl4
Ans 7: (a) Because phosphorous is the most electronegative element out of P, Bi, Sb and C.
Que 8. In the electrothermal process, the compound displaced by silica from calcium phosphate is
(a) Calcium (b) Phosphine
(c) Phosphorus (d) Phosphorus pentoxide
Ans 8: (d) Ca3(PO4)2 + SiO2 → CaSiO3 + P2O5
Que 9. Aqueous solutions of hydrogen sulphide and sulphur dioxide when mixed together, yield
(a) Sulphur and water (b) Sulphur trioxide and water
(c) Hydrogen peroxide and sulphur (d) Hydrogen and sulphurous acid
Ans 9: (a) SO2 + 2H2S → 3S + 2H2O
Que 10. An example of a neutral oxide is
(a) NO (b) CO2
(c) CaO (d) ZnO
Ans 10. (a)
Neutral oxides are those oxides that show neither basic nor acidic properties when they react with water. Examples include carbon monoxide (CO) and nitrous oxide (N2O) which are only slightly soluble in water, and nitric oxide (NO) which is appreciably soluble in cold water.
MCQs on The p-Block Elements Class 12 Chemistry
Que 11. Which of the following is the best scientific method to test the presence of water in a liquid
(a) Taste
(b) Smell
(c) Use of litmus paper
(d) Use of anhydrous copper sulphate 63. H2S react with O2 to form
(a) H2O + S (b) H2O + SO2
(c) H2O + SO3 (d) H2SO4 + S
Ans 11: a) 2H2S + O2 → 2H2O + 2S
Que 12. Which of the following mixture is chromic acid
(a) K2Cr2O7 and conc. H2SO4 (b) K2Cr2O7 and HCl
(c) K2SO4 and conc. H2SO4 (d) H2SO4 and HCl
Ans 12: (a) Mixture of K2Cr2O7 and conc. H2SO4 is known as chromic acid.
Que 13. The solubility of I2 increases in water in the presence of
(a) KI (b) H2SO4
(c) KMnO4 (d) NH3
Ans 13: (a) Iodine (I2 ) is slightly soluble in water but it dissolves in a 10% aqueous solution of KI due to the formation of potassium triiodide (KI3).
Que 14. Which of the hydrogen halides forms salts like KHX2 (where X is a halogen atom)
(a) HF (b) HCl
(c) HI (d) HBr
(e) All of these
Ans 14: (a) Due to the highest electronegativity of fluorine the anion [F – – – H – F]– exists as a result of a strong hydrogen bond by which K+ associate to form KHF2.
Que 15. With cold and dilute sodium hydroxide fluorine reacts to give
(a) NaF and OF2 (b) NaF + O3
(c) O2 and O3 (d) NaF + O2
Ans 15: (a) Fluorine is the most electronegative element. It does not form oxyfluoride like other halogens. It reacts with NaOH to form sodium fluoride and oxygen fluoride.
2NaOH + 2F2 → 2NaF + OF2 + H2O
MCQs on The p-Block Elements Class 12 Chemistry
Que 16. Which one of the following oxides is expected to exhibit paramagnetic behaviour
(a) CO2 (b) SO2
(c) ClO2 (d) SiO2
Ans 16: c) Due to unpaired e– ClO2 is paramagnetic.
Que 17. Of the following acids, the one that is strongest is
(a) HBrO4 (b) HOCl
(c) HNO2 (d) H3PO3
Ans 17: a) Oxidation number of HBrO4 is more than that of HOCl, HNO2 and H3PO3 so it is the strongest acid among these
Que 18. Which of the following is the anhydride of perchloric acid
(a) Cl2O7 (b) Cl2O5
(c) Cl2O3 (d) HClO
Ans 18: (a) Chlorine heptachloride (Cl2O7) is the anhydride of perchloric acid
Que 19. A tetra-atomic molecule (P) on reaction with nitrogen (I) oxide, produces two substances (Q) and (R). (Q) is a dehydrating agent in its monomeric form while substance (R) is a diatomic gas that shows almost inert behaviour. The substances (P) and (Q) and (R) respectively will be
(a) P4, N2O5, N2
(b) P4, P4O10, N2
(c) P4, P2O3, H2
(d) P4, P2O3, N2
Ans 19: (b). P4 + 10 NO → P4O10 + N2
Que 20. What may be expected to happen when phosphine gas is mixed with chlorine gas?
(a) The mixture only cools down
(b) PCl3 and HCl are formed and the mixture warms up
(c) PCl5 and HCl are formed and the mixture cools down
(d) PH3.Cl2 is formed with warming up
Ans 20: (b)
PH3 is highly poisonous it explodes with traces of oxidizing agents like HNO3, Cl2 and Br2 vapours.
PH3 + 3Cl2 → PCl3 + 3 HCl; ΔH = −ve
Hence, PCl3 and HCl are formed and the mixture warms up as heat is evolved.
MCQs on The p-Block Elements Class 12 Chemistry
Que 21. An orange solid (A) on heating gives a green residue (B), a colourless gas (C) and water vapours. The dry gas (C) on passing over heated magnesium gave a white solid (D). (D) on reaction with water gives a gas (E) which gives dense white fumes with HCl. Here (E) will be:
(a) N2O3 (b) NO3
(c) NH3 (d) N2O
Ans 21: (NH4)2Cr2O7 → Cr2O3 + N2 + 4H2O
(A) (B) (C)
N2 + 3 Mg → Mg3N2
D
Mg3N2 + 6 H2O → 3 Mg(OH)2 + 2 NH3
E
NH3 + HCl → NH4Cl (White Fumes)
Que 22. It is possible to obtain oxygen from air by fractional distillation because
(a) oxygen is in a different group of the periodic table from nitrogen
(b) oxygen is more reactive than nitrogen
(c) oxygen has higher b.p. than nitrogen
(d) oxygen has a lower density than nitrogen.
Ans 22. (c)
Air is liquified by making use of the joule-Thompson effect (cooling by the expansion of the gas) Water vapour and CO2 are removed by solidification. The remaining major constituents of liquid air i.e., liquid oxygen and liquid nitrogen are separated by means of fractional distillation (b.p. of O2 = –183°C: b. P. of N2 = – 195.8°C)
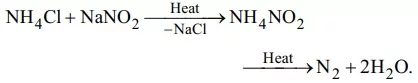
Que 23. Pure nitrogen is prepared in the laboratory by heating a mixture of
(a) NH4OH + NaCl (b) NH4NO3 + NaCl
(c) NH4Cl + NaOH (d) NH4Cl + NaNO2.
Ans 23: (d)
Que 24. On heating ammonium dichromate and barium azide separately we get
(a) N2 in both cases
(b) N2 with ammonium dichromate and NO with barium azide
(c) N2O with ammonium dichromate and N2 with barium azide
(d) N2O with ammonium dichromate and NO2 with barium azide
Ans 24: (a) N2 in both cases
Que 25. Oleum is
(a) Castor Oil (b) oil of vitriol
(c) fuming H2SO4 (d) None of them
Ans 25: (c)
Oleum is H2S2O7 (H2SO4 + SO3) which is obtained by dissolving SO3 in H2SO4 and is called fuming sulphuric acid.
Que 26. On addition of conc. H2SO4 to a chloride salt, colourless fumes are evolved but in the case of an iodide salt, violet fumes come out. This is because
(a) H2SO4 reduces HI to I2 (b) HI is of violet colour
(c) HI gets oxidised to I2 (d) HI changes to HIO3
Ans 26: (c) HI gets oxidised to I2
Que 27. Among the following which is the strongest oxidising agent?
(a) Br2 (b) I2
(c) Cl2 (d) F2
Ans 27: (d)
Since all the halogens have a strong tendency to accept electrons. Therefore, halogens act as strong oxidising agents and their oxidising power decreases from fluorine to iodine.
Que 28. The correct order of heat of formation of halogen acids is
(a) HI > HBr > HCl > HF (b) HF > HCl > HBr > HI
(c) HCl > HF > HBr > HI (d) HCl > HBr > HF > HI
Ans 28: (b)
On moving from top to bottom of halogen group the bond dissociation energy of hydrogen halides decreases and so the heat of formation of halogen acids also decreases.
Que 29. Which is the weakest out of HF, HCl, HBr and HI?
(a) HF (b) HCl (c) HBr (d) HI
Ans 29: (a)
Due to intermolecular H-bonding is weakest among HX acids
Que 30. Which of the following is correct about the reaction?
3 NaClO → NaClO3 + 2NaCl
(a) It is a disproportionation reaction
(b) Oxidation number of Cl decreases as well as increases in this reaction
(c) This reaction is used for the manufacture of halates
(d) All of these
Ans 30. (d)
+1 +5 -1
3 NaClO → NaClO3 + 2NaCl
MCQs on The p-Block Elements Class 12 Chemistry