Chlorine its Properties and Oxoacids of Chlorine
Chlorine (Cl2)
(1) Preparation: It can be prepared by any one of the following methods:
(i) By heating manganese dioxide with concentrated hydrochloric acid.
MnO2 + 4HCl → MnCl2 + Cl2 + 2H2O
Conc. HCl can be replaced by a mixture of common salt and concentrated H2SO4
4NaCl + MnO2 + 4H2SO4 → MnCl2+ 4NaHSO4 + 2H2O + Cl2
(ii) By the action of HCl on potassium permanganate.
2KMnO4 + 16HCl → 2KCl + 2MnCl2 + 8H2O + 5Cl2
(2) Manufacture of Chlorine:
(i) Deacon’s Process:
By oxidation of hydrogen chloride gas by atmospheric oxygen in the presence of CuCl2 (catalyst) at 723 K.
4HCl+O2 ⎯⎯⎯⎯→2Cl2 +2H2O
(ii) Electrolytic Process: Chlorine is obtained by the electrolysis of brine solution (concentrated NaCl solution). During electrolysis, chlorine is liberated at the anode.
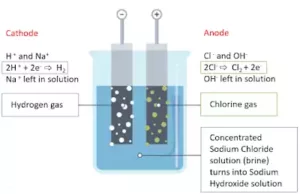
(3) Properties:
(i) It is a greenish-yellow gas with a pungent and suffocating odour.
(ii) It is soluble in water.
(iii) It reacts with a number of metals and non-metals to form chlorides.
2Al + 3Cl2 → 2AlCl3
P4 + 6Cl2 → 4PCl3
2Na + Cl2 → 2NaCl
S8 + 4Cl2 → 4S2Cl2
2Fe + 3Cl2 → 2FeCl3
(iv) With excess ammonia, chlorine gives nitrogen and ammonium chloride whereas with excess chlorine, nitrogen trichloride(explosive) is formed.
8NH3 + 3Cl2 → 6NH4Cl + N2
(excess)
NH3 + 3Cl2 → NCl3 + 3HCl
(excess)
(v) With cold and dilute alkalies chlorine produces a mixture of chloride and hypochlorite.
2NaOH + Cl2 → NaCl + NaOCl + H2O
(cold and dilute)
With hot and concentrated alkalies it gives chloride and chlorate.
6 NaOH + 3Cl2 → 5NaCl + NaClO3 + 3H2O
(hot and conc.)
(vi) With dry slaked lime it gives bleaching powder-+.
2Ca(OH)2 + 2Cl2 → CaOCl2 + CaCl2 + 2H2O
(vii) Chlorine reacts with hydrocarbons and gives substitution products with saturated hydrocarbons and addition products with unsaturated hydrocarbons.
CH4 + Cl2 ⎯⎯UV⎯→ CH3Cl + HCl
Methane Methyl chloride
(viii) Chlorine water on standing loses its yellow colour due to the formation of HCl and HOCl. Hypochlorous acid (HOCl)so formed is unstable and dissociates to give nascent oxygen which is responsible for oxidising and bleaching properties of chlorine.
(ix) It oxidises ferrous to ferric, sulphite to sulphate, sulphur dioxide to sulphuric acid and iodine to iodic acid.
2FeSO4 + H2SO4 + Cl2 → Fe2(SO4)3 + 2HCl
Na2SO3 + Cl2 + H2O → Na2SO4 + 2HCl
SO2 + 2H2O + Cl2 → H2SO4 + 2HCl
I2 + 6H2O + 5Cl2 → 2HIO3 + 10HCl
(x) It is a powerful bleaching agent. Bleaching action is due to oxidation.
Cl2 + H2O → 2HCl + [O]
Coloured substance + [O] → Colourless substance
It bleaches vegetable or organic matter in the presence of moisture. Its bleaching action is permanent.
(4) Uses of Cl2: It is used
(i) For bleaching wood pulp, bleaching cotton and textiles
(ii) In the extraction of gold and platinum
(iii) In the manufacture of dyes, drugs and organic compounds such as CCl4, CHCl3, DDT, refrigerants, etc.
(iv) In sterilizing drinking water
(v) Preparation of poisonous gases such as Phosgene (COCl2), Tear gas (CCl3NO2), Mustard gas (ClCH2CH2SCH2CH2Cl).
Chlorine its Properties and Oxoacids of Chlorine
Hydrochloric Acid (HCl)
(1) Preparation: It is prepared in the laboratory, by heating sodium chloride with concentrated sulphuric acid.
NaCl + H2SO4 ⎯⎯420⎯K⎯→ NaHSO4 + HCl
NaHSO4 + NaCl ⎯⎯823⎯K⎯→ Na2SO4 + HCl
(2) Properties:
(i) These are colourless, pungent-smelling gas with acidic tastes.
(ii) It is heavier than air, can be liquified to colourless liquids.
(iii) These are neither combustible nor supporter of combustion.
(iv) When perfectly dry, they have no action on litmus, but in presence of moisture, they turn blue litmus red, showing acidic nature. Among HX, HI is the strongest and HF is the weakest acid.
(v) These are quite soluble in water. HCl ionises as below.
HCl(g) + H2O (l) ® H3O+ (aq) + Cl– (aq) Ka = 107.
(vi) Its aqueous solution is called hydrochloric acid. The high value of the dissociation constant (Ka) indicates that it is a strong acid in water.
(vii) It reacts with NH3and gives white fumes of NH4Cl.
NH3 + HCl → NH4Cl
(viii) When three parts of concentrated HCl and one part of concentrated HNO3are mixed, aqua regia is formed which is used for dissolving noble metals, e.g., gold, platinum.
Au + 4 H+ + NO3– + 4Cl– →AuCl4– + NO + 2H2O
3Pt + 16H++ 4NO3– + 18Cl– → 3PtCl62- + 4NO + 8H2O
(ix) Hydrochloric acid decomposes salts of weaker acids like carbonates, hydrogen carbonates, sulphites, etc.
Na2CO3 + 2HCl → 2NaCl + H2O + CO2
NaHCO3 + HCl → NaCl + H2O + CO2
Na2SO3 + 2HCl → 2NaCl + H2O + SO2
(x) Reaction with metals oxides, hydroxides and bicarbonates
Zn + 2HCl → ZnCl2 + H2
MgO + 2HCl → MgCl2 + H2O
NaOH + HCl → NaCl + H2O
Detection of cation: HCl:
AgNO3 + HCl → AgCl¯ (white) + HNO3
(CH3COO)2Pb + 2HCl → PbCl2¯ (white) + 2CH3COOH
Hg(NO3)2 + 2HCl → Hg2Cl2 ¯ (white) + 2HNO3
(3) Uses: It is used
(i) In the manufacture of chlorine, NH4Cl and glucose (from corn starch).
(ii) For extracting glue from bones and purifying boneblack,
(iii) In medicine and as a laboratory
(iv) HI is used as a reducing agent in organic chemistry.
Chlorine its Properties and Oxoacids of Chlorine
Oxoacids of Halogens
The term oxoacid now refers to a compound that contains oxygen, at least one other element, and at least one hydrogen bound to oxygen.
(i) Fluorine forms only one oxoacid HOF (Fluoric (I) acid or hypofluorous acid)due to high electronegativity.
(ii) The other halogens form several oxoacids like Hypohalous acid (HOX), halous acid (HOXO), halic acid (HOXO2) and perhalic acid (HOXO3). They are stable only in aqueous solutions or in the form of their salts.
(iii) Chlorine forms 4 types of oxoacids –hypochlorous acid (HOCl), Chlorous acid (HOClO or HClO2), Choric acid (HOClO2 or HClO3) and perchloric acid (HOClO3 or HClO4). The structures of them are:
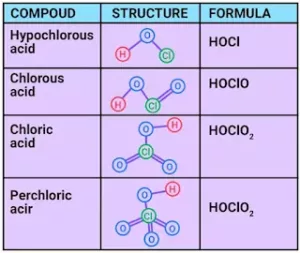
(iv) Acid strength: HOCl < HClO2< HClO3 < HClO4
Reason: HClO4 → H+ + ClO4– -most stable
(v) Acid strength: HOF > HOCl > HOBr > HOI
This is because Fluorine is the most electronegative element.
Chlorine its Properties and Oxoacids of Chlorine



