Case Study Questions p – Block Elements
Case Study Questions p – Block Elements
1. Read the passage given below and answer any four out of the following questions:
Ammonia is present in small quantities in air and soil where it is formed by the decay of nitrogenous organic matter e.g., urea. On a large scale, ammonia is manufactured by Haber’s process. In accordance with Le Chatelier’s principle, high pressure would favour the formation of ammonia. Ammonia is a colourless gas with a pungent odour. Its freezing and boiling points are 198.4 and 239.7 K respectively. In the solid and liquid states, it is associated with hydrogen bonds as in the case of water and that accounts for its higher melting and boiling points than expected on the basis of its molecular mass. Ammonia gas is highly soluble in water. Its aqueous solution is weakly basic due to the formation of OH– ions. The presence of a lone pair of electrons on the nitrogen atom of the ammonia molecule makes it a Lewis base.
1. The catalyst which is used in the preparation of NH3 by Haber’s process
(A) Zinc oxide, Mg2O3 + K2O
(B) Iron oxide, Al2O3 + K2O
(C) Copper oxide, Na2O + K2O
(D) Nickel oxide, CaO + K2O
Ans 1. (B) Iron oxide, Al2O3 + K2O
2. NH3 burns in the air under suitable conditions to produce
(A) N2
(B) NO
(C) N2O
(D) N9O3
Ans 2. (B) NO
3. A compound reacts with aqueous ammonia to form a deep blue colouration solution, identify the metal ion present in the compound.
(A) As2+
(B) Ca2+
(C) Cu2+
(D) Sb2+
Ans 3. (C) Cu2+
4. Which one of the following compounds on strong heating evolves ammonia gas?
(A) (NH4)2Cr2O7
(B) NH4NO3
(C) NH4NO2
(D) (NH4)2SO4
Ans 4. (NH4)2SO4
Case Study Questions p – Block Elements
2. Read the given passage and answer the questions that follow:
Group 16 elements are called chalcogens i.e., ore-forming elements (oxygen, sulphur, selenium etc.) because most of the ores are oxides and sulphides. Oxygen is gas whereas other elements of group 16 are solids. Oxygen shows anomalous behaviour. Oxygen is diatomic where is sulphur exists as S8 which has a crown-shaped structure. It shows allotropy. Sulphur is present in onion and garlic that is why they have a pungent smell. Sulphur is used for the manufacture of sulphuric acid which is called the ‘King of chemicals’, used in fertilizer, detergents, dyes and drugs.
1. Name the most abundant element in the earth crust.
Ans 1. Oxygen
2. A gas ‘X’ is obtained from the roasting of sulphide ore. It turns lime water milky. Identify the gas and write the chemical reaction involved.
Ans 2. ‘X’ is SO2. It turns lime water milky due to the formation of calcium sulphite.
Ca(OH)2 + SO2(g) → CaSO3(s) + H2O(l)
3. What happens when SO2 gas is reacted with O2 in presence of V2O5 at 770K temperature and high pressure? Write the chemical reaction involved.
Ans 3. SO3 gas is formed.
4. What happens when SO3 is passed through sulphuric acid? Write the chemical equation for the reaction.
Ans 4. Oleum is formed, SO
SO3 + H2SO4 → H2S2O7
5. Why is SO3 not directly absorbed in water to get sulphuric acid?
Ans 5. It is because the reaction is highly exothermic, beyond control.
Case Study Questions p – Block Elements
3. Read the passage given below and answer the following questions:
The halogens have the smallest atomic radii in their respective periods. The atomic radius of F is extremely small. All halogens exhibit a -1 oxidation state. They are strong oxidizing agents and have maximum negative electron gain enthalpy. Among halogens, F shows anomalous behaviour in many properties. For example, electronegativity and ionization enthalpy are higher for F than expected whereas bond dissociation enthalpy, m.p and b.p and electron gain enthalpy are quite lower than expected. Halogens react with hydrogen to give hydrogen halides (HX) and combine amongst themselves to form a number of compounds of type XX’, XX’3, XX’5, XX’7 called interhalogens.
1. The stability of interhalogen compounds follows the order
(A) IF3 > BrF3 > ClF3
(B) ClF3 > BrF3 > IF3
(C) BrF3 > IF3 > ClF3
(D) ClF3 > IF3 > BrF3
Ans 1. (A) IF3 > BrF3 > ClF3
2. Identify the correct match from the following
(A) [ICl2] – Bent
(B) IF7 – Pentagonal bipyramidal
(C) ClF3 – Trigonal planar
(D) [BrF4]– –- Square pyramidal
Ans 2. (B) IF7 – Pentagonal bipyramidal
3. In XX’5, the central atom has
(A) 5 bond pairs and 0 lone pair
(B) 5 bond pairs and one lone pair
(C) 6 bond pairs and 0 lone pair
(D) 4 bond pairs and one lone pair
Ans 3. (B) 5 bond pairs and one lone pair
4. Which of the following is not the characteristic of interhalogen compounds?
(A) They are more reactive than halogens.
(B) They are quite unstable but none of them is explosive.
(C) They are covalent in nature.
(D) They have low boiling points and are highly volatile.
Ans 4. (D) They have low boiling points and are highly volatile.
5. Which of the following statements are correct?
(A) Among halogens, the radius ratio between iodine and fluorine is maximum.
(B) Leaving F – F bond, all halogens have weaker X – X bond than X – X’ bond in interhalogens.
(C) Among inter-halogen compounds maximum number of atoms at present in iodine (III) fluoride.
(D) Inter-halogen compounds are more reactive than halogen compounds
Ans 5. (C) Among inter-halogen compounds maximum number of atoms at present in iodine (III) fluoride.
Case Study Questions p – Block Elements
4. Read the following passage and answer the questions that follow:
Group 18 elements are called noble gases and not inert gases because compounds of Kr, Xe and Rn have been prepared. Their general electronic configuration is ns2np6 except He(1s2). They have the highest ionisation enthalpy and positive electron gain enthalpy due to stable electronic configuration. Helium is found in the sun and stars. Noble gases have low boiling points due to weak van der Waals’ forces of attraction. Xenon forms XeF2, XeF4, XeF6, XeOF4, XeO3, XeO2F2, their structures can be drawn on bases of VSEPR theory. Helium is mixed with oxygen by deep-sea divers to avoid pain. Neon is used in coloured advertising lights. Argon is used in bulbs as an inert gas. Kr and Xe are used in high-efficiency lamps, headlights of cars. Radon is radioactive formed by a-decay of Radium 88226Ra Argon is the most abundant (0.9%) noble gas in the atmosphere.
1. How does boiling points of noble gases vary down the group?
Ans 1. The boiling point of noble gases increases down the group due to an increase in atomic size, surface area and an increase in van der Waals’ forces of attraction.
2. Which noble gas has the highest ionisation enthalpy and why?
Ans 2. Helium. It is due to the smallest atomic size.
3. Which was the first noble gas compound prepared by Neil Bartlett?
Ans 3. Xe+PtF6–
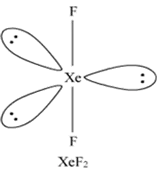
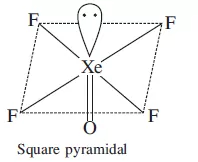
4. Draw the shapes of
(i) XeF2
(ii) XeOF4.
Ans 4. (i)
(ii)
5. Complete the following reactions:
(i) XeF6 + KF →
(ii) XeF6 + 2H2O →
Ans 5. (i) XeF6 + KF → K+[XeF7]–
(ii) XeF6 + 2H2O → XeO2F2 + 4HF
Case Study Questions p – Block Elements