London Dispersion Forces Examples
London Dispersion Forces Examples
Intermolecular forces are the forces of attraction and repulsion between interacting particles (atoms and molecules). Attractive intermolecular forces are known as van der Waals forces. These forces include dispersion forces or London forces, dipole-dipole forces, and dipole-induced dipole forces. A particularly strong type of dipole-dipole interaction is hydrogen bonding.
In the year 1930, a scientist Fritz London once explained that noble gas atoms have the ability to attract another atom by some forces such forces named as London forces.
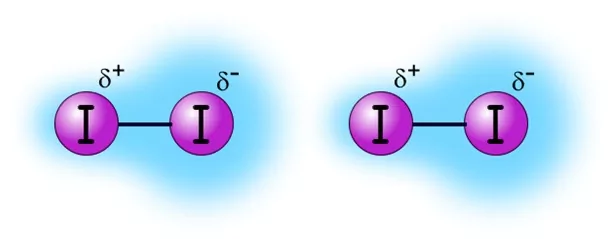
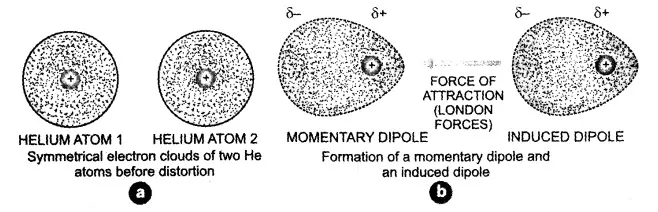
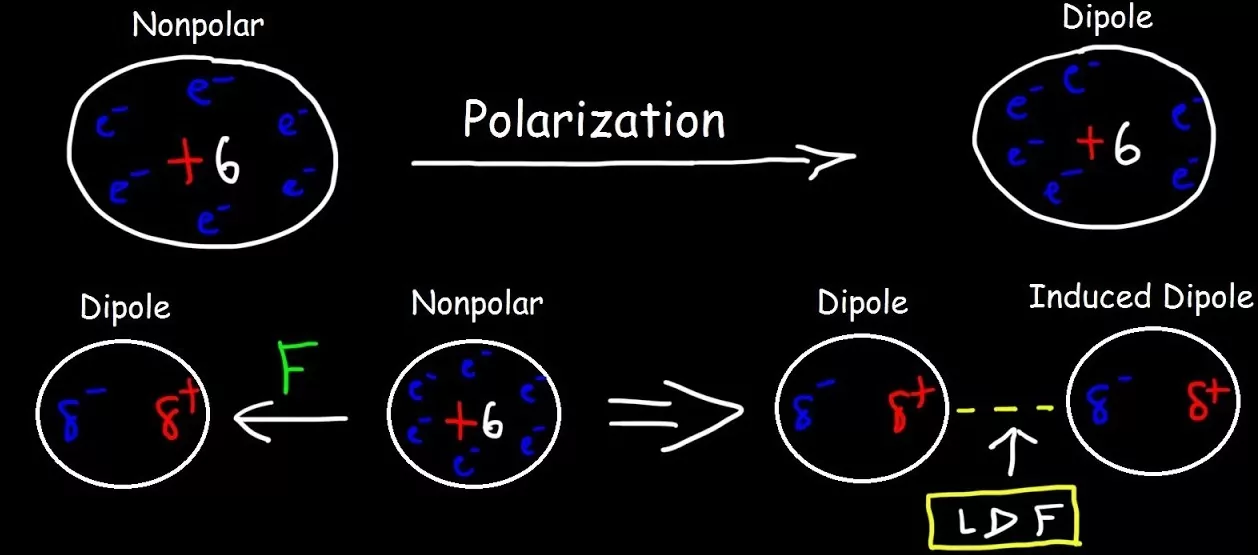
The London dispersion force is the weakest temporary attractive intermolecular force. London dispersion forces can explain how liquids and solids form in non-polar molecules. “Dispersion” means the way things are distributed or spread out. The London dispersion force is a force that results when the electrons in two adjacent atoms occupy positions that make the atoms form temporary dipoles. These temporary dipoles of atoms attract each other. This force of attraction between temporary dipoles is termed as London forces or dispersion forces. These forces are important only at short distances.
London Dispersion Forces in I2 molecule
London Dispersion Forces Examples
Forces of attraction between momentary dipoles and induced dipoles (London forces) in Helium atoms
Dispersion forces are present between all molecules, whether they are polar or nonpolar.
(i) Larger and heavier atoms and molecules exhibit stronger dispersion forces than smaller and lighter ones.
(ii) In a larger atom or molecule, the valence electrons are, on average, farther from the nuclei than in a smaller atom or molecule. They are less tightly held and can more easily form temporary dipoles.
(iii) The ease with which the electron distribution around an atom or molecule can be distorted is called Polarizability.
London dispersion forces tend to be:
(i) Stronger between molecules that are easily polarized.
(ii) Weaker between molecules that are not easily polarized.
London Dispersion Forces Examples