Properties of Ionic Bond and Octet Rule
Properties of Ionic Bond and Octet Rule
It is observed that the atoms of all the elements, except those for noble gases, tend to remain in a combined state with the atoms of the same or other elements. They do not exist as single atoms under ordinary conditions. Such atomic aggregates occur as molecules.
| The attractive force that binds the atoms together in a molecule is called a chemical bond. It is formed either by the transfer of electrons or by the sharing of electrons. |
The elements with one, two, three, four, five, six, or seven electrons in the outer shell, use these electrons to complete octet. The electrons which take part in two or more atoms to complete an octet is known as Electrovalency.
Lewis Dot Structure: It is a shorthand to represent the valence electrons of an atom. The structures are written as the element symbol surrounded by dots that represent the valence electrons. The inner electrons are well protected and they are called core electrons.
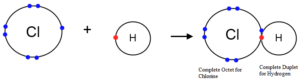
 Octet Rule: This rule was proposed by Lewis and Kossel. According to this rule, atoms undergo chemical reactions in order to attain an octet of electrons in the valence shell. Or, atoms containing 8 electrons in their valence shell are stable. To attain octet, they share electrons or exchange electrons. Furthermore, the atom does this by bonding with each other. Moreover, these atoms can be the same element or with different elements.
Octet Rule: This rule was proposed by Lewis and Kossel. According to this rule, atoms undergo chemical reactions in order to attain an octet of electrons in the valence shell. Or, atoms containing 8 electrons in their valence shell are stable. To attain octet, they share electrons or exchange electrons. Furthermore, the atom does this by bonding with each other. Moreover, these atoms can be the same element or with different elements.
Two notable exceptions to the octet rule are helium and hydrogen. This is because both are happy with two electrons belonging to the outer shells.

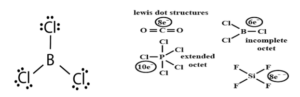
Limitations of octet rule:
Formation of electron-deficient compounds like BeCl2, BF3, AlCl3. Central atom has less than 8e–.
Formation of hypervalent compounds like PCl5, SF6, IF7, H2SO4 in which the central atom has more than 8 electrons.
Formation of compounds of noble gases like XeF2, XeF4, XeF6.
Odd electron molecules like NO, NO2.
Types of Chemical Bonds: The chemical bonds are classified into the following types
- Ionic bond
- Covalent bond
- Dative bond or Co-ordinate bond
- Hydrogen bond
- Metallic bond
Properties of Ionic Bond and Octet Rule
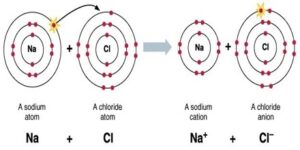
Ionic bond: A bond formed by the transfer of an electron between 2 atoms is called an ionic or electrovalent bond. Or, it is the force of attraction between two oppositely charged ions. These ions are held together by the electrostatic force of attraction. The number of electrons lost or gained is known as the electro-valency.
Na → Na+ +e–
2,8,1 2,8
Cl + e– → Cl–
2,8,7 2,8,8
Na+ + Cl– → NaCl
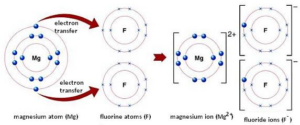
Let us take another example of MgCl2 (Magnesium Chloride):
Energy changes during the formation of an Ionic Bond:
An ionic bond formation involves the following steps:
The formation of a positive ion (cation) from the electropositive atom. The amount of energy required for this process is called ionization enthalpy (∆iH).
M(g) + ∆iH → M+(g) + e–
Formation of negative ion from the electronegative atom. The energy involved in this process is called electron gain enthalpy (∆egH).
X(g) + e– → X–(g) + ∆egH
The packing of the cation and anion to form an ionic compound. The energy change in this process is called lattice enthalpy (∆latticeH)
M+ + X– → MX(s) + ∆latticeH
Factors favouring the formation of ionic bond:
Low ionization enthalpy (IE)
More negative electron gain enthalpy (EGE)
High lattice enthalpy (LE) If LE + EGE > IE, an ionic bond is formed.
Properties of ionic compounds:
Non-directional nature of ionic bond: These compounds do not exist as individual molecules. Instead, they are often hard solids, always made up of ions held together by strong electrostatic forces of attraction between ions with opposite charge.
Electrical conductivity: When added to water, the ions separate. The solution can thus conduct electricity. Similarly, these compounds conduct electricity in the fused state also.
Melting and boiling points: These compounds generally have high melting points. This is because a lot of thermal energy is required to break down the inter-ionic forces and form a liquid. Similarly, they possess high boiling points.
Solubility: These compounds are usually soluble in water but insoluble in benzene or other organic solvents.
Properties of Ionic Bond and Octet Rule
Covalent Bond: When a bond is formed by the sharing of electrons by the two atoms the bond formed between them is called a covalent bond. Each pair of shared electrons is indicated by a line -. Once the covalent bond has been formed, the two bonding electrons are attracted by the two nuclei instead of one, so the bonded state becomes more stable than the non-bonded state.
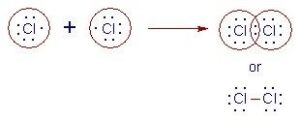
For eg. The formation of a Chlorine molecule (Cl2)
17Cl + 17Cl → Cl2
2,8,7 2,8,7
Both the chlorine atoms have 7 electrons in their outermost shell. So here the exchange of electrons is not possible. For the formation of a Cl2 molecule, each Cl atom shares one pair of electron. The bond thus formed is called a single covalent bond.
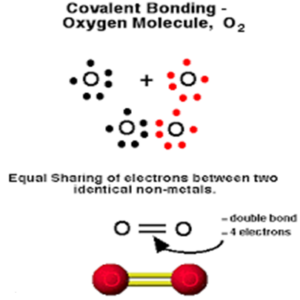
The formation of an O2 molecule
8O + 8O → O2
2,6 2,6
To attain an octet, each oxygen atom shares 2 pairs of electrons. Thus, a double covalent bond is formed.
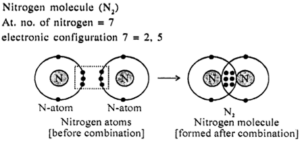
The formation of N2 molecule
7N + 7N → N2
2,5 2,5
To attain the octet configuration, the nitrogen atoms share 3 pairs of electrons. Thus a triple bond is formed.
The number of covalent bonds formed by an atom is termed as its covalency. In the formation of the Cl2 molecule, the covalency of each Cl atoms is one, in the O2 molecule, the covalency of O atom is 2 and in N2, it is 3. The double and triple bonds are together called multiple bonds.
Properties of Covalent Compounds: The covalent compounds are found to possess the following general properties, which are opposite to those of ionic compounds:
(a) These substances are made up of individual covalent molecules with weak intermolecular forces.
(b) Because of the weak intermolecular forces, the substances are gases, liquids, or soft solids at room temperature.
(c) These substances when put into the water do not undergo ionization. Hence these substances in the fused state or in solution do not conduct electricity. However, in presence of water, some polar covalent compounds can ionize and conduct electricity.
(d) These substances have low melting and boiling points because less energy is needed to break down the weak intermolecular forces.
(e) These substances are commonly soluble in benzene and other organic solvents but insoluble in water.
https://sciencemotive.com/class11/resonance-dipole-moment-and-bond-parameters/
Properties of Ionic Bond and Octet Rule



