Hydrocarbon MCQ for NEET PDF – Class 11 Chemistry
Hydrocarbon MCQ for NEET PDF
Que 1. Which of the following is Baeyer’s reagent?
(a) Alkaline KMnO4
(b) Acidic K2Cr2O7
(c) Alkaline Na2Cr2O7
(d) MnO2
Ans 1. (a)
Baeyer’s reagent is an alkaline solution of cold potassium permanganate (KMnO4).
Que 2. The function of anhydrous AlCl3 in Friedel– Crafts reaction is
(a) To absorb water
(b) To absorb HCl
(c) To produce attacking electrophile
(d) To produce nucleophile.
Ans 2. (c)
AlCl3 produces attacking electrophile
Cl2 + AlCl3 → Cl+ + AlCl4–
Que 3. Meta-directing and deactivating group in aromatic electrophilic substitution is
(a) – CH3
(b) – OH
(c) – NO2
(d) – Cl
Ans 3. (c)
– CH3: Activating and o, p-directing
– OH: Activating and o, p-directing
– NO2: Deactivating and m-directing
– Cl: Deactivating and o, p-directing
Que 4. The numbers of s and p-bonds present in 1, 3-butadiene are respectively
(a) 9 and 2
(b) 8 and 2
(c) 9 and 3
(d) 9 and 1
Ans 4. (a)

Que 5. Which of the following reaction is not correct?
(a) Propene + HCl → 2-chloropropane
(b) Propene + HBr → 1-bromopropane
(c) Propene + HCl → 2-chloropropane
(d) Propene + HBr → 1-bromopropane
Ans 5. (d)
CH3 ̶ CH = CH2 + HCl → CH3 ̶ CH(Cl) ̶ CH3
Que 6. Arrange the following hydrogen halides in order of their decreasing reactivity with propene.
(a) HCl > HBr > HI
(b) HBr > HI > HCl
(c) HI > HBr > HCl
(d) HCl > HI > HBr
Ans 6. (c)
The decreasing order of reactivity of halo acids with propene is HI > HBr > HCl. As the size of halogen increases, the strength of H–X bond decreases, and hence, reactivity increases
Que 7. The IUPAC name of the compound having the formula CH ≡ C – CH = CH2 is
(a) 1-butyn-2-ene
(b) but-1-yn-3-ene
(c) But-1-en–3-yne
(d) 3-buten-1-yne
Ans 7. (c) But-1-en–3-yne
Que 8. Anti-Markownikov addition of HBr is not observed in
(a) propene
(b) 1-butene
(c) 2-butene
(d) 2-pentene
Ans 8. (c)
Anti-Markownikov addition of HBr is observed only with unsymmetrical alkenes i.e., propene, 1-butene, 2-pentene. As, 2-butene is symmetrical so in its case Anti – Markownikov addition will not be observed.
Que 9. Mark the correct decreasing order of stability.
(a) Aromatic > non-aromatic > anti-aromatic
(b) Aromatic > anti-aromatic > non-aromatic
(c) Non-aromatic > anti-aromatic > aromatic
(d) Anti-aromatic > non-aromatic > aromatic
Ans 9. (a)
Aromatic is most stable followed by non-aromatic and anti-aromatics are least stable.
Que 10. Which among the following is a very strong o-, p-directing group?
(a) ̶ Cl
(b) ̶ OR
(c) ̶ NH2
(d) ̶ NHR
Ans 10. (d) The directive influence order is
O– > NR2 > NHR > NH2 > OH > OCH3 ≈ NHCOCH3 > CH3 > X
Que 11. Electrolysis of an aqueous solution of sodium ethanoate gives
(a) methane
(b) ethane
(c) butane
(d) methyl ethanoate.
Ans 11. (b)
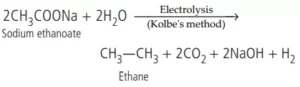
Que 12. Which of the following compounds reacts with sodium to liberate hydrogen gas?
(a) Ethane
(b) Propene
(c) Acetylene
(d) Benzene
Ans 12. (c)
Only acetylene has acidic hydrogens and hence reacts with Na to evolve H2 gas.
![]()
Que 13. The correct order of reactivity towards electrophilic substitution is
(a) phenol > benzene > chlorobenzene > benzoic acid
(b) benzoic acid > chlorobenzene > benzene > phenol
(c) phenol > chlorobenzene > benzene > benzoic acid
(d) benzoic acid > phenol > benzene > chlorobenzene
Ans 13. (a)
In general electron-releasing groups, activate and electron-withdrawing groups deactivate the benzene ring towards electrophilic substitutions. Thus the order of reactivity is:
Phenol > Benzene > Chlorobenzene > Benzoic acid
Que 14. Which of the following compounds will not undergo Friedel-Crafts reaction easily?
(a) Nitrobenzene
(b) Toluene
(c) Cumene
(d) Xylene
Ans 14. (a)
Nitrobenzene is strongly deactivated, hence will not undergo Friedel-Crafts reaction.
Que 15. Benzene easily shows
(a) ring fission reactions since it is unstable
(b) addition reactions since it is unsaturated
(c) electrophilic substitution reactions due to stable ring and high p electron density
(d) nucleophilic substitution reactions due to stable ring and minimum electron density.
Ans 15. (c)
The most common reactions shown by benzene are electrophilic substitution reactions.
(a) Assertion and reason both are correct statements and reason is the correct explanation for assertion.
(b) Assertion and reason both are correct statements but the reason is not the correct explanation for assertion.
(c) Assertion is a correct statement but the reason is a wrong statement.
(d) Assertion is the wrong statement but the reason is a correct statement.
Que 16. Assertion: The boiling point of alkanes increases with an increase in molecular weight.
Reason: van der Waal’s forces increase with an increase in molecular weight.
Ans 16. (a)
Greater is the molecular mass, greater is the magnitude of van der Waal’s forces of attraction, and hence higher the boiling point.
Que 17. Assertion: Acetylene is acidic in nature.
Reason: Acetylene is sp hybridized.
Ans 17. (b)
The acidic nature in acetylene is described on the basis of the higher electronegativity of sp hybridized carbon atom which pulls the C – H bond pair more effectively to lose H+.
Que 18. Assertion: Trans-pent-2-ene is polar but trans-but-2-ene is non-polar.
Reason: The polarity of cis-isomer is more than trans which are either non-polar or less polar.
Ans 18. (b)
The vector sum of all polar bonds in trans-pent-2- ene is not zero but the vector sum is zero in trans-but-2-ene.
Que 19. Give two reactions to show the acidic character of alkynes.
Ans 19. CH ≡ CH + Na → HC ≡ C– Na+ + 1/2 H2
CH ≡ CH + NaNH2 → HC ≡ C–Na+ + NH3
Hydrocarbon MCQ for NEET PDF



