Surface Chemistry Class 12 NCERT Solutions Que 5.1. Distinguish between the meaning of the terms adsorption and absorption. Give one example of each. Ans 5.1. Difference between adsorption and absorption S.No. Adsorption Absorption 1 The phenomenon of accumulation of the molecules of a substance on a solid or liquid surface In absorption, the substance is uniformly distributed throughout the body of a solid or a liquid. 2. It is a surface phenomenon. It is a bulk phenomenon. 3. Here, the concentration of the substance increases on the surface. Here, the concentration of the substance increases in the bulk. 4.…
Author: Dr. Vikas Jasrotia
Surface Chemistry NCERT Intext Solutions Que 1. Write any two characteristics of Chemisorption. Ans 1. Two characteristics of Chemisorption- (i) The bond formation between the solid(adsorbent) and the gas molecules is by chemical bonds. (ii) It is highly specific in nature and also irreversible. Que 2. Why does physisorption decrease with the increase of temperature? Ans 2. Physical adsorption of a gas by a solid is generally reversible. Thus, Solid + Gas ↔ Gas / Solid + Heat Since the adsorption process is exothermic, the physical adsorption occurs readily at low temperatures and decreases with increasing temperatures (Le Chatelier’s…
Electrochemistry Class 12 NCERT Exercise Solutions Que 1. Arrange the following metals in the order in which they displace each other from their salts. Al, Cu, Fe, Mg, and Zn Ans 1. The order in which metals displace each other from the solution of their salts can be given with the help of their standard electrode potential. Since magnesium has the least standard electrode potential so it is the most strong reducing agent. So the required order we get is Mg, Al, Zn, Fe, Cu Que 2. Given the standard electrode potentials, K+/K=-2. 93 V, Ag+/Ag = 0.80 V, Hg2+/Hg =…
NCERT Intext Questions Class 12 Electrochemistry Que 1. How would you determine the standard electrode potential of the system Mg2+|Mg? Ans 1. We know that it is not possible to measure the electrode potential of a half cell. We can only measure the difference between electrode potentials of two half cells, which gives the cell potential of the cell. For measuring the standard electrode potential of the given system, we need to use Mg as one electrode and a standard hydrogen electrode as the second electrode. Since the electrode potential of the standard hydrogen electrode is zero, the value of…
Important Assertion Reason Questions of Electrochemistry Que 1. Assertion: In an electrochemical cell anode and cathode are respectively negative and positive electrodes. Reason: At anode oxidation takes place and at cathode reduction takes place. Ans 1. (a) Both assertion and reason are true and the reason is the correct explanation of assertion. Explanation: Anode (oxidation) for eq Zn → Zn2+ + e- Excess of electrons and hence negatively charged while the cathode is positively charged. Que 2. Assertion: Galvanised iron does not rust. Reason: Zn has more (–) ve electrode potential than Fe. Ans 2. (a) Que 3. Assertion: Conductivity…
Chapter – 2 Chapter Solutions Assertion Reason Questions In the following questions, a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choice. (a) Assertion and reason both are correct statements but the reason is the correct explanation for assertion. (b) Assertion and reason both are correct statements but the reason is not a correct explanation for assertion. (c) Assertion is a correct statement but the reason is a wrong statement. (d) Assertion is a wrong statement but the reason is correct Assertion Reason Questions Que 1. Assertion: Ice…
Assertion Reason Questions Solid State In the following questions, two statements Assertion and Reason are given. Mark (a) Both Assertion and Reason are correct and Reason is the correct explanation of Assertion. (b) Both Assertion and Reason are correct but Reason is not the correct explanation of Assertion. (c) Assertion is true but Reason is false. (d) Assertion is false but Reason is true. (e) Assertion and Reason both are false. —————————————————————————————————— Que 1. Assertion: For fluorite structure, the F– ions occupy tetrahedral void, but Ca+2 ions occupy c.c.p. Reason: The radius ratio of fluorite structure is 0.225. Ans 1.…
Coordination Compounds NEET Questions Que 1. In K4[Fe(CN)6] (a) (CN) are linked with primary valency (b) (CN) are linked with secondary valency (c) K is linked with secondary valency (d) K is linked with non-ionic valency Ans 1. (b) (CN) are linked with secondary valency Explanation: (CN) are linked with secondary valency to the central Fe metal ion. Ligand (CN) is linked to the central atom via a dative bond. Que 2. In the coordination compound, K4[Ni(CN)4], the oxidation state of nickel is (a) 0 …
Coordination Compounds Important Questions Que 1. (i) Write down the IUPAC name of the following complex: [Pt(NH3)(H2O)Cl2] and K3[Fe(CN)6] (ii) Write the formula for the following complex: tris(ethane-l,2-diamine)chromium(III) chloride and Pentaaminenitrito-N-cobalt(III) Ans 1. (i) Ammineaquadichloridoplatinum (II) and Potassiumhexacyanoferrate(III) ion (ii) [Cr(en)3]Cl3 and [CO(NH3)5NO2]2+ Que 2. Ni(CO)4 possesses tetrahedral geometry, while [Pt(NH3)4]2+ is square planar. Why? Ans 2. Ni(CO)4 possesses sp3 hybridization and then tetrahedral, whereas [Pt(NH3)4]2+ possesses dsp2 hybridization, thus square planar. Que 3. Explain the following: (i) NH3 act as a ligand but NH4+ does not. (ii) CN− is a ambidentate ligand. Ans. (i) NH3 has one lone pair…
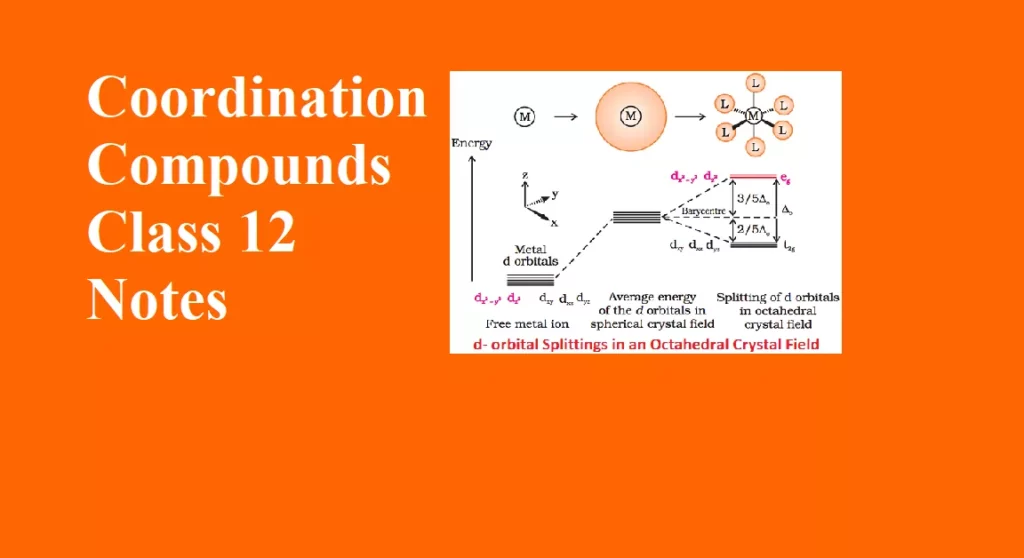
Coordination Compounds Class 12 Notes Important Topics The concept of coordination compounds originates from the tendency for the complex formation of the transition elements. Complex compounds or coordination compounds are those molecular compounds that retain their identity in solid and in solution are known as complex compounds. K4[Fe(CN)6 ] + H2O → 4K+(aq) + [Fe(CN)6]4− (aq) Molecular or Addition Compounds: When solutions of two or more simple stable compounds in molecular proportion are allowed to evaporate, crystals of new substances, called molecular or addition compounds, are obtained. For Example: CuSO4 + 4NH3 → CuSO4·4NH3 AgCN + KCN → KCN·AgCN…