Amines Important Questions Class 12 – Chemistry
Amines Important Questions
Que 1. Why do amines act as Lewis Base?
Ans 1. Amines have an unshared pair of electrons on nitrogen atoms due to which they behave as lewis base.
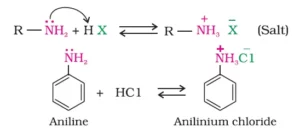
Que 2. Which diazonium salt is stable at room temperature?
Ans 2. Benzene diazonium floro borate(C6H5N2 F).
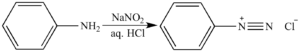
Que 3. What is diazotization? Write an example.
Ans 3. The conversion of primary aromatic amines into diazonium salts is known as diazotization.
Que 4. Which one is more basic CH3NH2 or (CH3)3N in the gaseous phase and why?
Ans 4. (CH3)3N is more basic because the greater number of alkyl groups increases the magnitude of the +I effect so increases the basicity.
Que 5. Why is an alkylamine more basic than ammonia?
Ans 5. Due to the electron releasing inductive effect (+I) of an alkyl group, the electron density on the nitrogen atom increases, and thus, it can donate the lone pair of electrons more easily than ammonia.
Que 6. Out of Butan-1-ol and butan-1-amine, which will be more soluble in water and why?
Ans 6. Butan-1-ol are more polar than amines and forms stronger intermolecular hydrogen bonds with water molecules than amines.
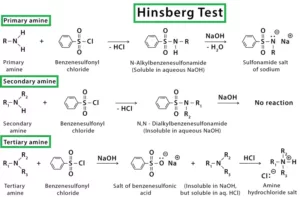
Que 7. Describe Hinsberg’s test for the identification of primary, secondary, and tertiary amines. Also, write the chemical equations of the reactions involved.
Ans 7. Hinsberg’s test: Benzenesulphonyl chloride (C6H5SO2Cl), commonly known as Hinsberg’s reagent, forms sulphonamides when it interacts with primary and secondary amines.
(i). For Primary Amines: N-ethylbenzenesulphonyl amide is produced by reacting benzenesulphonyl chloride with a primary amine. Because of the strong electron-withdrawing sulphonyl group, the hydrogen linked to nitrogen in sulphonamide is highly acidic. As a result, it is alkali-soluble.
(ii). For Secondary Amines: N,N-diethylbenzenesulphonamide is produced in the reaction with a secondary amine. It is not acidic and hence insoluble in alkali because N, N-diethylbenzene sulphonamide has no hydrogen atom linked to the nitrogen atom.
(iii).For Tertiary Amines: Benzenesulphonyl chloride does not react with tertiary amines. This characteristic of amines reacting differently with benzenesulphonyl chloride is used to distinguish primary, secondary, and tertiary amines, as well as to separate a mixture of amines.
Amines Important Questions
Que 8. Out of CH3NH2 and CH3CH2NH2 which has a higher boiling point and why?
Ans 8. CH3CH2NH2 because the bigger the alkyl group more is the surface area higher is the magnitude of van der Waals force.
Que 9. Arrange the following in the increasing order of given property indicated
(i). C2H5NH2, (C2H5)2NH, (C2H5)3N, and NH3, (Basic strength in aqueous solution).
(ii). C2H5NH2, (C2H5)2NH, (C2H5)3N, and NH3, (Basic strength in gaseous phase).
(iii). Aniline, p-toluidine, p-nitroaniline. (Basic strength).
Ans 9 (i). The increasing order of basic strength in aqueous solution is:
NH3 < C2H5NH2 < (C2H5)3N < (C2H5)2NH
(ii). The increasing order of basic strength in gaseous phase is:
NH3 < C2H5NH2 < (C2H5)2NH < (C2H5)3N
(iii). The increasing order of basic strength of the compounds is given as:
p-nitroaniline < Aniline < p-toluidine
Que 10. Why Ethylamine is soluble in water whereas aniline is not?
Ans 10. Ethylamine when added to water forms intermolecular H−bonds with water. Hence, it is soluble in water.
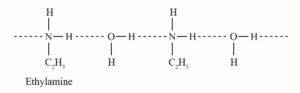
But aniline Can form H−bonding with water to a very small extent due to the presence of a large hydrophobic −C6H5 group. Hence, aniline is insoluble in water.
Que 11. Give a chemical test to distinguish between ethylamine and aniline. (All India 2011)
Ans 11. Ethylamine and aniline :
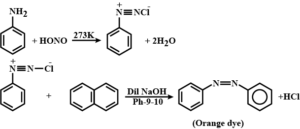
By Azo dye test: It involves the reaction of any aromatic primary amine with HNO2(NaNO2 + dil. HCl) at 273-278 K followed by treatment with an alkaline solution of 2-naphthol when a brilliant yellow, orange or red-colored dye is obtained.
Que 12. Answer the following questions:
(i) The quaternary ammonium salts having four different alkyl groups are optically active.
(ii) Alkylamines are more basic than ammonia.
(iii) Aniline cannot be prepared by Gabriel phthalimide synthesis.
(iv) Gabriel phthalimide synthesis is preferably used for synthesizing primary amines.
Ans 12. (i). There is no rapid inversion in quaternary ammonium compounds because nitrogen atoms do not have a lone pair of electrons. The optical activity of the nitrogen atom is due to its sigma bonding to four alkyl groups.
(ii). The alkyl group (R) pulls electrons towards nitrogen in alkyl amine due to its electron-releasing nature, making the unshared electron pair more available for sharing with the acid’s proton. Therefore, alkyl amines show a more basic nature than ammonia.
(iii). Because aryl halides cannot undergo nucleophilic substitution with the anion produced by phthalimide, Gabriel’s phthalimide synthesis technique cannot produce aniline.
(iv). This process is used to make primary amines from primary alkyl halides. Because secondary and tertiary amines are not generated during this synthesis, only pure primary amines are produced, which is why this reaction is favored for primary amine production. N-Alkyl Phthalimide is formed as a result of this reaction.
Que 13. Aniline does not undergo Friedel-Crafts reaction. Why?
Ans 13. Because the reagent AlCl3 (the Lewis acid used as a catalyst in Friedel crafts reactions) is electron deficient, it functions as a Lewis base and does not undergo Friedel craft reactions. It attacks the lone pair of nitrogen in aniline, forming an insoluble complex that precipitates out, stopping the process. That is why aniline does not undergo Friedel-Crafts reaction.
Amines Important Questions



