Case Study Questions Chapter 11 Chemistry
1. Read the passage given below and answer the following questions
Alcohols are some of the most important molecules in organic chemistry. Alcohols contain the hydroxy functional group (-OH), bonded to a carbon atom of an alkyl or a substituted alkyl group. The electronegativity of oxygen is substantially greater than that of carbon and hydrogen. Consequently, the covalent bonds of this functional group are polarized so that oxygen is electron-rich and both carbon and hydrogen are electrophilic. Indeed, the dipolar nature of the O–H bond is such that alcohols are much stronger acids than alkanes (by roughly 1030 times), and nearly that much stronger than ethers. The most reactive site in an alcohol molecule is the hydroxyl group, despite the fact that the O–H bond strength is significantly greater than that of the C–C, C–H, and C–O bonds, demonstrating again the difference between thermodynamic and chemical stability.
Q 1. Which one is more acidic?
(a) C2H6 (b) CH4 (c) CH3OCH3 (d) C2H5-OH
Ans 1. (d) C2H5-OH
Q 2. The acidic character of alcohols is due to
(a) Capability of forming H-bonding
(b) Polar nature of O-H bond
(c) -I effect of an alkyl group
(d) None of these
Ans 2. (b) Polar nature of O-H bond
Q 3. The acidic strength of alcohols decreases in the following order
(a) 1° > 2° > 3°
(b) 3° > 2° > 1°
(c) 2° > 1° > 3o
(d) 3° > 1° >2°
Ans 3. (a) 1° > 2° > 3°
Case Study Questions Chapter 11 Chemistry
2. Read the given passage and answer the questions that follow:
Alcohols and phenols are the most important compounds used in our daily life. Alcohols are prepared by hydration of alkenes, fermentation of glucose, reduction of aldehydes, ketones, carboxylic acids, and esters.
Alcohols are soluble in water. Boiling points increase with an increase in molar mass and decrease with branching. Alcohols on dehydration give alkene at 443K, follow the carbocation mechanism. Excess of alcohol at 413K on dehydration with conc. H2SO4 also follows a carbocation mechanism but gives diethyl ether. Alcohols undergo nucleophilic substitution reactions, esterification with carboxylic acids and derivatives like amides, acid halides, acid anhydride Phenol is prepared from cumene, diazonium salts, anisole, chlorobenzene. Phenol is used to prepare salicylaldehyde, salicylic acid, aspirin, methyl salicylate, p-benzoquinone. Phenol undergoes electrophilic substitution reaction at o & p-position. Ethers are functional isomers of alcohols, have low boiling points. Ethers are used as solvents. Unsymmetrical ethers are prepared by Williamson synthesis. Ethers react with HI and undergo SN1 or SN2 mechanisms depending upon the stability of carbocation formed. Aromatic ethers like anisole undergo electrophilic substitution at o & p – position.
Answer the following questions:
Q 1. The IUPAC name of
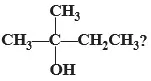
(a) 2-methyl butanol
(b) 2-methyl butan-2-ol
(c) Methyl butan-2-ol
(d) 2-methyl propan-2-ol
Ans 1. (b) 2-methyl butan-2-ol
Q 2. Out of tert, butyl alcohol, and n-butanol, which will undergo dehydration faster?
(a) tert, butyl alcohol
(b) n-butanol
(c) Both have the same tendency
(d) None undergo dehydration
Ans 2. Tertiary butyl alcohol will undergo dehydration faster because 3° carbocation is more stable.
Q 3. Complete the reaction
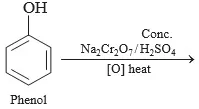
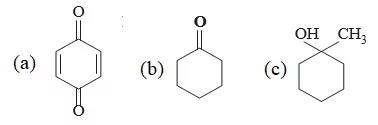
Ans 3. (a)
Q 4. Why is C—OH bond length in CH3OH longer than C—OH bond length in phenol?
Ans 4. It is due to resonance in phenol, there is a double bond character in C=O bond, therefore shorter than the single bond in methanol.
Case Study Questions Chapter 11 Chemistry
3. Read the passage given below and answer the following questions
The facility with which the aromatic ring of phenols and phenol ethers undergoes electrophilic substitution has been noted. Two examples are shown in the following diagram. The first shows the Friedel-Crafts synthesis of the food preservative BHT from para-cresol. The second reaction is interesting in that it further demonstrates the delocalization of charge that occurs in the phenolate anion. Carbon dioxide is a weak electrophile and normally does not react with aromatic compounds; however, the negative charge concentration on the phenolate ring enables the carboxylation reaction shown in the second step. The sodium salt of salicylic acid is the major product, and the preference for ortho substitution may reflect the influence of the sodium cation. This is called the Kolbe-Schmidt reaction, and it has served in the preparation of aspirin, as the last step illustrates.
Q 1. IUPAC name of salicylic acid
(a) o-Hydroxybenzoic acid
(b) p- Hydroxybenzoic acid
(c) 2- Hydroxybenzoic acid
(d) 3- Hydroxybenzoic acid
Ans 1. (c) 2- Hydroxybenzoic acid
Q 2. Salicylic acid heated with acetic anhydride to form a compound which is very useful drug which lower the body temperature as well as use to prevent heart attack. What is the name of compound?
(a) 3-Acetyl Salicylic acid
(b) 2-Acetyl Salicylic acid
(c) 2-Acetoxybenzoic acid
(d) 3-Acetoxybenzoic acid
Ans 2. (c) 2-Acetoxybenzoic acid
Q 3. Salicylic acid is heated with acetic anhydride to form a compound. In this reaction which bond is cleavage in Salicylic acid?
(a) O-H bond of -COOH group
(b) O-O bond of -COOH group
(c) C-O bond of benzene ring and phenolic group
(d) O-H bond of phenolic group
Ans 3. (d) O-H bond of phenolic group
Case Study Questions Chapter 11 Chemistry



