Alcohols Phenols and Ethers Case-Based Questions
Alcohols Phenols and Ethers Case-Based Questions
1. Read the given passage and answer the questions that follow:
Alcohols play a very important role in our daily life. Ordinary spirit used as an antiseptic contains methanol. Ethanol is present in cough syrups, tonics, wine, beer, and whisky, Sugar, starch, cellulose are carbohydrates that also contain a large number —OH groups. Phenol is also an antiseptic in low concentration (0.2%) whereas a 2% solution of phenol is used as a disinfectant. The fragrance of rose is due to citronellol (unsaturated alcohol). Phenol is used for the preparation of many useful compounds like aspirin, methyl salicylate (Iodex), and phenyl salicylate (salol) used as an intestinal antiseptic.
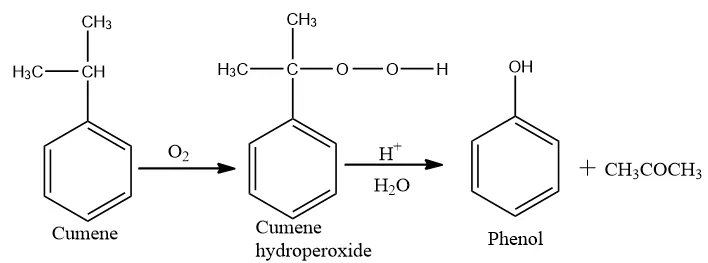
(a) How is phenol prepared from cumene? What is the advantage of this method?
Ans (a). Phenol is industrially prepared by the ‘Dow’s Process‘.
This process involves the following steps:
(i) Cumene is oxidized in presence of air at 400K in the presence of a metal catalyst to form cumene hydroperoxide.
(ii) In the second step cumene hydroperoxide is treated with dilute sulphuric acid at 350K. It causes hydrolysis and forms phenol and acetone.
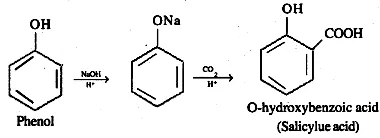
(b) How is phenol converted into salicylic acid?
Ans (b). Phenol is converted to salicylic acid by using Kolbe’s reaction. In this reaction the carboxyl group can be introduced directly into a phenol nucleus by passing carbon dioxide over the heated sodium phenoxide was first observed by Kolbe. Substitution occurs predominantly in the ortho position.
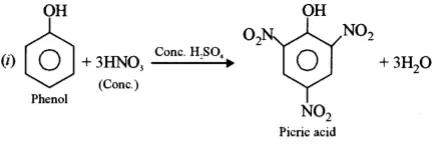
(c) Convert phenol to picric acid.
Ans (c). It can be converted using the following reaction
(d) Distinguish between phenol and benzyl alcohol?
Ans (d). Add neutral FeCl3. Phenol gives violet colour whereas benzyl alcohol does not.
(e) Why does phenol turn pink after long-standing?
Ans (e). It is due to oxidation.
2. Read the passage given below and answer the following questions:
The reaction of phenol with aqueous sodium hydroxide indicates that phenols are stronger acids than alcohols and water. Due to the higher electronegativity of sp2 hybridized carbon of phenol to which –OH is attached, electron density decreases on oxygen. This increases the polarity of the O–H bond and results in an increase in ionization of phenols than that of alcohols. Now let us examine the stabilities of alkoxide and phenoxide ions. In alkoxide ion, the negative charge is localized on oxygen while in phenoxide ion, the charge is delocalized. The delocalisation of negative charge makes phenoxide ions more stable and favours the ionization of phenol.
1. Phenol is less acidic than_________.
(A) Ethanol (B) o-nitrophenol (C) o-methylphenol (D) o-methoxy phenol
Ans 1. (B)
Explanation: Phenol is less acidic than o-nitrophenol as the electron-withdrawing (−NO2) group increases the acidity of phenols
2. Which of the following is most acidic?
(A) Benzyl alcohol (B) Cyclohexanol (C) Phenol (D) m-Chlorophenol
Ans 2. (D)
Explanation: m-chlorophenol is most acidic as the electron-withdrawing (−Cl) group increases the acidity of phenols
3. Phenol can be distinguished from ethanol by the reaction with _____
(A) Br2/water (B) Na (C) Glycerol (D) All of the above
Ans 3. (A)
Explanation: Phenol decolorizes bromine water to form a white precipitate of 2,4,6-tribromophenol whereas ethanol does not precipitate.